Advertisements
Advertisements
प्रश्न
Explain the following, giving two examples:
Coordination polyhedron
Explain with two examples of the following:
Coordination polyhedron
Advertisements
उत्तर
A coordination polyhedron is defined by the spatial arrangement of the ligand atoms that are directly connected to the centre atom or ion. For example:
- [Cu(NH3)4]2+: The coordination polyhedron of copper in this complex is a square planar geometry, as there are four ammonia ligands arranged in a square around the central copper ion (coordination number 4).
- [Fe(CN)6]4−: The coordination polyhedron of iron in this complex is octahedral, with six cyanide ligands arranged around the central iron ion (coordination number 6).
APPEARS IN
संबंधित प्रश्न
Write the structure and IUPAC names of all the metamers represented by formula C4H10
What is meant by unidentate ligand?
Which of the following compounds is used as a semipermeable membrane?
(a) Potassium ferrocyanide
(b) Potassium argentocyanide
(c) Sodium meta aluminate
(d) Copper ferrocyanide
IUPAC name of the following compound is
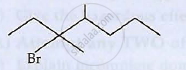
(a) 3 - Bromo- 3, 4- dimethylheptane
(b) 3, 4- dimethyl - 3- bromoheptane
(c) 5- Bromo- 4, 5- dimethylheptane
(d) 4, 5- dimethyl- 5- bromoheptane
Following compounds are given to you :
2-Bromopentane, 2-Bromo-2-methylbutane, 1-Bromopentane
1) Write the compound which is most reactive towards SN2 reaction.
2) Write the compound which is optically active.
3) Write the compound which is most reactive towards β-elimination reaction.
Which of the following represents a chelate ligand?
Ligand (en) is an example of ___________.
The coordination number of Cr in [Cr(NH3)3(H2O)3]Cl3 is ___________.
What is the coordination number of chromium in \[\ce{[Cr(NH3)2(H2O2)2]Cl3}\]?
Which of the following is non-ionizable?
Which of the following complexes formed by \[\ce{Cu^2+}\] ions is most stable?
The stabilisation of coordination compounds due to chelation is called the chelate effect. Which of the following is the most stable complex species?
A coordination compound \[\ce{CrCl3.4H2O}\] precipitates silver chloride when treated with silver nitrate. The molar conductance of its solution corresponds to a total of two ions. Write structural formula of the compound and name it.
Assertion: \[\ce{Cr(H2O)6]Cl2 and [Fe(H2O)6]Cl2}\] are reducing in nature.
Reason: Unpaired electrons are present in their d-orbitals.
The co-ordinate number and the oxidation state of the element E in the complex [E(en)2(C2O4)]NO2 are respectively?
The oxidation number d-arbitral occupation and co-ordination number of Cr in the complex cis [Cr(en)2Cl2]Cl are respectively.
Which of the following ligands can exhibit linkage isomerism?
What are Homoleptic complexes?
Why chelate complexes are more stable than complexes with unidentate ligands?
Explain the following, giving two examples:
Coordination number
