Advertisements
Advertisements
प्रश्न
Explain the following, giving two examples:
Coordination polyhedron
Explain with two examples of the following:
Coordination polyhedron
Advertisements
उत्तर
A coordination polyhedron is defined by the spatial arrangement of the ligand atoms that are directly connected to the centre atom or ion. For example:
- [Cu(NH3)4]2+: The coordination polyhedron of copper in this complex is a square planar geometry, as there are four ammonia ligands arranged in a square around the central copper ion (coordination number 4).
- [Fe(CN)6]4−: The coordination polyhedron of iron in this complex is octahedral, with six cyanide ligands arranged around the central iron ion (coordination number 6).
APPEARS IN
संबंधित प्रश्न
Classify the following ligands into monodentate and polydentate —
- Ammonia
- Carbon monoxide
- Ethylene diamine
- Ethylene diamine tetra acetate ion
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
cis-[CrCl2(en)2]Cl
Predict the co-ordination No. of cs+ ion if `r_(Cs)^+` = 1.69Å and `r_(Cl)^-` = 1.81Å.
Write the structures of compounds A, B and C in the following reactions
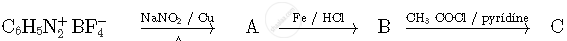
A group of atoms can function as a ligand only when:
The coordination number of the central ion may be obtained from:
In which of the following compounds the oxidation state of the nickel atom is 0?
Arrange the following complexes in the increasing order of conductivity of their solution:
[Co(NH3)3Cl3], [Co(NH3)4Cl2]Cl, [Co(NH3)6]Cl3, [Cr(NH3)5Cl]Cl2
A coordination compound \[\ce{CrCl3.4H2O}\] precipitates silver chloride when treated with silver nitrate. The molar conductance of its solution corresponds to a total of two ions. Write structural formula of the compound and name it.
Match the complex ions given in Column I with the colours given in Column II and assign the correct code:
| Column I (Complex ion) | Column II (Colour) |
| A. \[\ce{[Co(NH3)6]^{3+}}\] | 1. Violet |
| B. \[\ce{[Ti(H2O)6]^{3+}}\] | 2. Green |
| C. \[\ce{[Ni(H2O)6]^{2+}}\] | 3. Pale blue |
| D. \[\ce{(Ni(H2O)4 (en)]^{2+} (aq)}\] | 4. Yellowish orange |
| 5. Blue |
What are Homoleptic complexes?
Why chelate complexes are more stable than complexes with unidentate ligands?
Given below are two statements.
Statement I: In CuSO4·5H2O, Cu-O bonds are present.
Statement II: In CuSO4·5H2O, ligands coordinating with Cu(II) ion are O- and S-based ligands.
In light of the above statement, choose the correct answer from the options given below.
The following ligand is:

The one that will show optical activity is: (en = ethane 1, 2-diamine)
What is meant by the chelate effect? Give an example.
Explain the ligand.
Give two examples of ambidentate ligands.
