Advertisements
Advertisements
प्रश्न
Explain the formation of [Co(NH3)6]3+ ion on the basis of valence bond theory.
Based on valence bond theory, predict the magnetic behaviour of the [Co(NH3)6]3+ complex.
स्पष्ट कीजिए
विस्तार में उत्तर
Advertisements
उत्तर
- The oxidation state of Cobalt is +3.
- The valence shell electronic configuration of Co3+ is represented in the box diagram, as shown below:
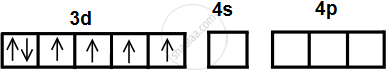
- The number of ammine ligands is 6, and the number of vacant metal ion orbitals required for bonding with ligands must be six.
- Complexes are low-spin, so pairing of electrons will take place prior to hybridisation.
- Electronic configuration after pairing would be
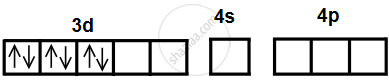
- Six orbitals available for hybridisation are two 3d, one 4s, three 4p orbitals.
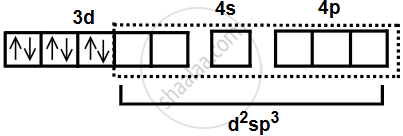
The orbitals for hybridization are decided from the number of ammine ligands, which is six. Here, (n − 1)d orbitals participate in hybridization since it is the low-spin complex. - Electronic configuration after complex formation.
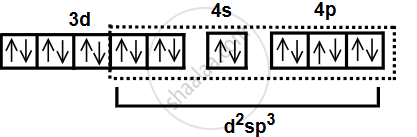
- According to Valence Bond Theory (VBT), the Co3+ ion undergoes d2sp3 hybridization in an octahedral field.
- As all electrons are paired, the complex is diamagnetic.
- If there are unpaired electrons, the complex is paramagnetic.
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
