Advertisements
Advertisements
प्रश्न
Give an example of chelate effect.
अति संक्षिप्त उत्तर
Advertisements
उत्तर १
\[\ce{Ni^{2+}_{ (aq)} + 6NH_{3 (aq)} <-> \underset{log \beta = 7.99}{[Ni(NH3)6]}^{2+}_{ (aq)}}\]
\[\ce{Ni^{2+}_{ (aq)} + 3en_{(aq)} <-> \underset{(more stable)}{\underset{log \beta = 18.1}{[Ni(en)_3]}}^{2+}_{ (aq)}}\]
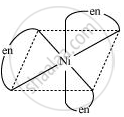
shaalaa.com
उत्तर २

or, \[\ce{[PtCl2(en)]}\]
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
अध्याय 5: Coordinate Compounds - Exercises [पृष्ठ १३९]
