Advertisements
Advertisements
Question
Give an example of chelate effect.
Very Short Answer
Advertisements
Solution 1
\[\ce{Ni^{2+}_{ (aq)} + 6NH_{3 (aq)} <-> \underset{log \beta = 7.99}{[Ni(NH3)6]}^{2+}_{ (aq)}}\]
\[\ce{Ni^{2+}_{ (aq)} + 3en_{(aq)} <-> \underset{(more stable)}{\underset{log \beta = 18.1}{[Ni(en)_3]}}^{2+}_{ (aq)}}\]
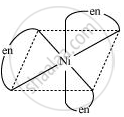
shaalaa.com
Solution 2

or, \[\ce{[PtCl2(en)]}\]
shaalaa.com
Notes
Students can refer to the provided solutions based on their preferred marks.
Is there an error in this question or solution?
Chapter 5: Coordinate Compounds - Exercises [Page 139]
