Advertisements
Advertisements
प्रश्न
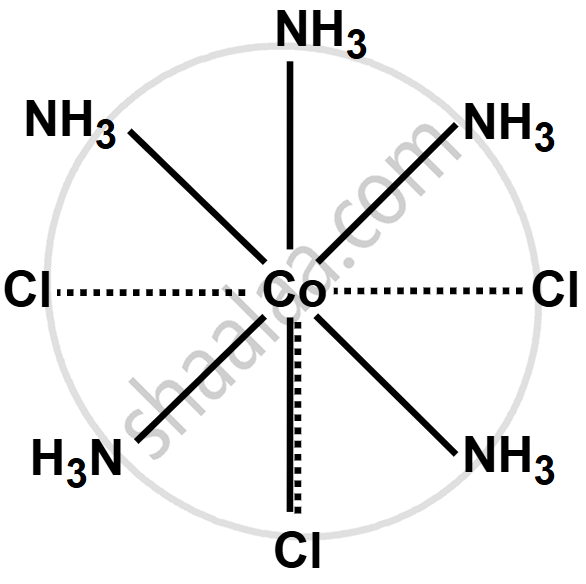
Write the structure of the following complex on the basis of Werner’s theory:
CoCl3·5NH3
रासायनिक समीकरण/संरचनाएँ
Advertisements
उत्तर
The oxidation state of cobalt in the complex CoCl3·5NH3 is +3. This gives three primary valencies, with three Cl atoms bonded to cobalt by these valencies. The remaining five NH₃ molecules are attached by secondary valencies, making the coordination number of cobalt six. One Cl atom is linked by both primary and secondary valencies and does not ionize in solution. The complex ionizes as follows:
\[\ce{CoCl3.5NH3 -> [CoCl.5NH3]^2+ + 2Cl^-}\]
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
