Advertisements
Advertisements
प्रश्न
Write the geometrical isomers of the complex [PtBrCl(NH3)2].
दीर्घउत्तर
Advertisements
उत्तर
- The complex [PtBrCl(NH3)2] exhibits geometrical isomerism due to its square planar geometry and the different types of ligands attached to the central platinum ion.
- In the cis-isomer, the two like ligands, the ammonia (NH3) molecules, occupy adjacent positions (90° apart), while the halide ligands (Br− and Cl−) are also adjacent.
- This leads to the formation of the cis-form of the complex.
- In contrast, in the trans-isomer, the two NH3 ligands are positioned opposite each other (180° apart), as are the Br− and Cl− ligands.
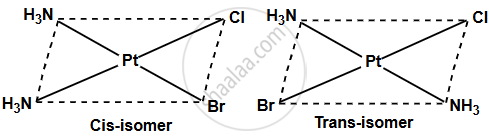
Therefore, the complex [PtBrCl(NH3)2] exists in two geometrical isomers: the cis-isomer and the trans-isomer, each having a distinct arrangement of ligands around the platinum ion.
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
