Advertisements
Advertisements
प्रश्न
What is meant by monodentate ligands? Illustrate with examples.
विस्तार में उत्तर
Advertisements
उत्तर
The ligands which possess only one donor atom and can form only one coordinate bond to the central metal atom or ion are called unidentate ligands or monodentate ligands. Example of monodentate ligands:
- Ammonia (NH3): Ammonia is a monodentate ligand because the nitrogen atom donates a lone pair of electrons to the metal ion.
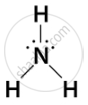
In the complex [Cu(NH3)4]2+ ammonia acts as a monodentate ligand by donating its lone pair to the copper ion. - Chloride ion (Cl−): The chloride ion (Cl−) is a monodentate ligand because it has a lone pair of electrons on the chlorine atom that can be donated to the metal ion.

In the complex [AgCl2]− chloride ions act as monodentate ligands by bonding to the silver ion.
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
