Advertisements
Advertisements
प्रश्न
How does the presence of a particular ligand affect the thermodynamic stability of a complex? Illustrate your answer with a suitable example.
Advertisements
उत्तर
The presence of a particular ligand plays a crucial role in determining the thermodynamic stability of a complex. Ligands influence stability through factors such as their field strength, charge, size, and ability to chelate. Here’s how these factors work:
1. Nature of the Ligand (Field Strength):
- Strong field ligands (like CN−, CO, NH3) form stronger bonds with the central metal ion, leading to higher stability of the complex.
- Weak field ligands (like Cl−, F−, H2O) form weaker bonds, resulting in lower stability of the complex.
2. Chelation Effect:
- Chelating ligands (ligands that can form more than one bond to the metal center) significantly increase the stability of the complex. This is due to the formation of cyclic structures.
- For example, EDTA (a hexadentate ligand) forms extremely stable complexes with metals because it can bind to the metal at multiple sites.
3. Charge on the Ligand:
- Ligands with higher negative charges (e.g., CN−, OH−) generally increase the electrostatic attraction between the metal and the ligand, enhancing the stability of the complex.
- Neutral ligands like NH3 form relatively less stable complexes compared to negatively charged ligands like CO or CN−.
4. Size and Shape of the Ligand:
- Smaller ligands like NH3 or CO allow for a closer approach to the metal ion, leading to more stable complexes.
- Larger ligands might sterically hinder the complexation process, potentially lowering the thermodynamic stability.
5. Ligand Exchange:
- Some ligands are easier to replace than others, affecting the overall stability. For instance, NH3 is harder to displace compared to Cl−, making complexes with NH₃ more stable.
Example of a ligand affecting the thermodynamic stability of a complex:
- [Ni(CO)4]: CO is a strong field ligand that donates electron density to the metal and also accepts electron density via π-back donation. This interaction leads to a very stable complex. Additionally, CO is a neutral ligand, which further minimizes repulsion and increases the stability of the complex.
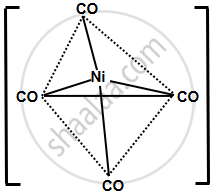
- [NiCl4]2−: Cl− is a weak field ligand, and its interaction with Ni2+ is relatively weak. The electrostatic repulsion between the negatively charged Cl− ions also weakens the bond with the metal, leading to lower stability in comparison to the CO complex.
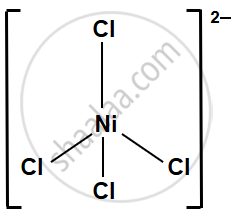
The complex [Ni(CO)4] is much more stable than [NiCl4]2− because CO is a strong field ligand and Cl− is a weak field ligand. Therefore, the nature of the ligand plays a crucial role in determining the stability of a complex.
