Advertisements
Advertisements
प्रश्न
Draw the structure and write the hybridisation state of the central atom of the following specie:
[Co(NH3)6]3+
रासायनिक समीकरण/संरचनाएँ
Advertisements
उत्तर
- The central atom is Cobalt (Co) in the +3 oxidation state, coordinated to six ammonia (NH3) ligands.
- The complex has an octahedral geometry because of the six ligands surrounding the central metal ion, each bonding with a lone pair of electrons.
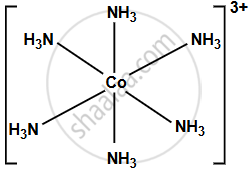
- The central Co3+ ion undergoes d2sp3 hybridisation.
- The hybridisation involves two d orbitals, one s orbital, and three p orbitals from cobalt, which combine to form six hybrid orbitals, each overlapping with a lone pair of electrons from each NH3 ligand.
- The geometry resulting from this hybridisation is octahedral.
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
