Advertisements
Advertisements
प्रश्न
Using the valence bond approach, predict the shape, hybridisation and magnetic behaviour of [Ni(CO)4]. (at. no. of Ni = 28)
Advertisements
उत्तर १

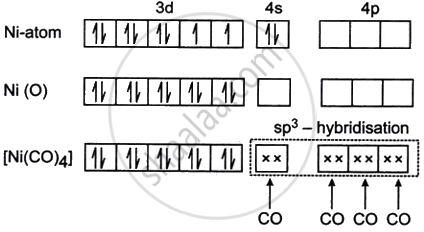
- The atomic number of nickel is 28, and its ground-state electronic configuration is [Ar] 3d84s2.
- In the complex [Ni(CO)4], nickel is in the zero oxidation state because CO is a neutral ligand.
- The electronic configuration of Ni in this state is 3d84s2.
- Carbon monoxide (CO) is a strong field ligand according to the spectrochemical series.
- Being a strong field ligand, CO causes the pairing of electrons in the 3d orbitals of nickel.
- As a result, the 3d orbitals become filled, while the 4s and 4p orbitals remain vacant.
- The vacant orbitals (one 4s and three 4p) undergo hybridisation to form four equivalent sp3 hybrid orbitals.
- Each of these hybrid orbitals accepts a lone pair of electrons from CO ligands through coordinate bonds.
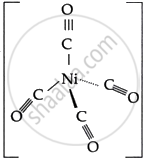
- Due to sp3 hybridisation, the geometry of the complex is tetrahedral.
- All the 3d electrons are paired, the complex has no unpaired electrons.
Therefore, [Ni(CO)4] is diamagnetic.
उत्तर २
Since CO is a neutral ligand, the oxidation state of Ni in [Ni(CO)4] is 0. Ni has an oxidation state of 0 in [Ni(CO)4], and its configuration is 3d84S2. As the electrons pair together inside the orbital, making CO a strong field ligand. As a result, the ligand-CO’s incoming electron pairs fill the remaining ‘s’ and ‘p’ orbitals, filling all five d-orbitals (2 in ‘s’ and 6 in ‘p’). Since there are four CO atoms in the complex, -CO fills the one’s orbital and the three ‘p’ orbitals. Because there are no unpaired electrons, the hybridization is sp3, and the geometry is tetrahedral, making it diamagnetic.