ISC (Science)
Academic Year: 2019-2020
Date: मार्च 2020
Advertisements
All questions are compulsory
- Question 1 is of 20 marks having four sub parts, all of which are compulsory.
- Question numbers 2 to 8 carry 2 marks each, with two questions having internal choice.
- Question numbers 9 to 15 carry 3 marks each, with two questions having an internal choice.
- Question numbers 16 to 18 carry 5 marks each, with an internal choice.
- All working, including rough work, should be done on the same sheet as, and adjacent to the rest of the answer.
- The intended marks for questions or parts of questions are given in brackets [ ].
- Balanced equations must be given wherever possible and diagrams where they are helpful.
- When solving numerical problems, all essential working must be shown.
- In working out problems, use the following data:
Gas constant R = 1.987 cal deg−1 mol−1 = 8.314 J K−1 mol−1 = 0.0821 dm3 atm K−1 mol−1
1 l atm = 1 dm3 atm = 101.3 J, 1 Faraday = 96500 coulombs
Avogadro’s number = 6.023 × 1023
Fill in the blanks by choosing the appropriate word/words from those given in the brackets:
(iodoform, volume, mass, haloform, gram equivalent, chloroform, carbylamine, sp3d2, high, coke, d2sp3, low, gram mole, carbon monoxide)
Equivalent conductivity is the conducting power of all the ions furnished by one ______ of an electrolyte present in a definite ______ of the solution.
Chapter:
Fill in the blanks by choosing the appropriate word/words from those given in the brackets:
(iodoform, volume, mass, haloform, gram equivalent, chloroform, carbylamine, sp3d2, high, coke, d2sp3, low, gram mole, carbon monoxide)
Bleaching powder, on treatment with ethanol or acetone gives ______. This is an example of ______ reaction.
Chapter:
Fill in the blanks by choosing the appropriate word/words from those given in the brackets:
(iodoform, volume, mass, haloform, gram equivalent, chloroform, carbylamine, sp3d2, high, coke, d2sp3, low, gram mole, carbon monoxide)
Outer orbital complexes involve ______ hybridization and are ______ spin complexes.
Chapter:
Fill in the blanks by choosing the appropriate word/words from those given in the brackets:
(iodoform, volume, mass, haloform, gram equivalent, chloroform, carbylamine, sp3d2, high, coke, d2sp3, low, gram mole, carbon monoxide)
Zinc oxide is reduced by ______ at 1673 K to form zinc and ______.
Chapter:
The packing efficiency of simple cubic structure, body centred cubic structure and face-centred cubic structure respectively is ______.
52.4%, 74%, 68%
74%, 68%, 52.4%
52.4%, 68%, 74%
68%, 74%, 52.4%
Chapter:
When acetone is treated with Grignard’s reagent, followed by hydrolysis, the product formed is ______.
Secondary alcohol
Tertiary alcohol
Primary alcohol
Aldehyde
Chapter:
Which of the following electrolytes is least effective in causing the flocculation of positively charged ferric hydroxide sol?
K3[Fe(CN)6]
K2CrO4
K4[Fe(CN)6]
KBr
Chapter:
On heating an aliphatic primary amine with chloroform and alcoholic potassium hydroxide, the organic compound formed is an ______.
Alkyl isocyanide
Alkanol
Alkanal
Alkyl cyanide
Chapter:
Match the following:
| (i) | Silicon and phosphorous | (a) | Acetaldehyde |
| (ii) | Iodoform test | (b) | Xenon hexafluoride |
| (iii) | Arrhenius equation | (c) | n-type of semiconductors |
| (iv) | Distorted octahedral structure | (d) | Frequency factor |
Chapter:
What is the common name of the polymer obtained by the polymerization of caprolactam? Is it an addition polymer or condensation polymer?
Chapter:
Why Zn2+ ions are colourless while Ni2+ ions are green and Cu2+ ions are blue in colour?
Chapter:
Calculate the molar conductivity at infinite dilution for CH3COOH if the molar conductivity at infinite dilution for NaCl, HCl and CH3COONa are 126.45, 426.16 and 91.0 ohm−1 cm2 mol−1 respectively.
Chapter:
Identify the compounds A, B, C and D.
\[\ce{C6H5COOH ->[SOCl2] A ->[NH3] B ->[Br2/KOH] C ->[NaNO2 + HCl][0-5^\circ C] D}\]
Chapter:
An element has atomic mass 93 g mol−1 and density 11.5 g cm–3. If the edge length of its unit cell is 300 pm, identify the type of unit cell. (NA = 6.023 × 1023 mol−1)
Chapter:
Calculate the radius of copper atom. The atomic weight of copper is 63.55 g mol−1. It crystallises in face-centred cubic lattice and has density of 8.93 g cm−3 at 298 K.
(NA = 6.023 × 1023 mol−1)
Chapter:
Complete and balance the following chemical equation:
\[\ce{P4 + NaOH + H2O ->[Heat][Inert atm.]}\] ______ + ______
Chapter:
Complete and balance the following chemical equation:
\[\ce{Cu + \underset{dil.}{HNO3} ->}\] ______ + ______ + ______
Chapter:
Write the chemical equation for the reaction of glucose with bromine water.
Chapter:
Advertisements
Name a substance which can be used as an antiseptic as well as disinfectant.
Chapter:
An alloy of gold (Au) and cadmium (Cd) crystallises with a cubic structure in which gold atoms occupy the corners and cadmium atoms fit into the face centres. What is the formula of this alloy?
Chapter:
Account for the following.
Ethylamine is soluble in water whereas aniline is not.
Chapter:
State reasons for the following:
Aliphatic amines are stronger bases than aromatic amines.
Chapter:
Complete and balance the following equation:
\[\ce{C6H5NH2 + CH3COCl ->}\] ______ + ______
Chapter:
Complete and balance the following equation:
\[\ce{C2H5NH2 + HNO2 ->}\] ______ + ______ + ______
Chapter:
Draw the structure of xenon tetrafluoride molecule and state the hybridization of the central atom and the geometry of the molecule.
Chapter:
Calculate the emf and ΔG for the given cell at 25°C:
Cr(s) | Cr3+ (0.1 M) || Fe2+ (0.01 M) || Fe(s)
Given: \[\ce{E^{\circ}_{Cr^{3+}/Cr}}\] = −0.74 V, \[\ce{E^{\circ}_{Fe^{2+}/Fe}}\] = −0.44 V
(1 F = 96500 C mol−1, R = 8.314 JK−1 mol−1)
Chapter:
Calculate the degree of dissociation (α) of acetic acid if its molar conductivity (Λm) is 39.05 S cm2 mol−1.
(Given \[\ce{\lambda^{\circ}_{(H^+)}}\] = 349.6 S cm2 mol−1 and \[\ce{\lambda^{\circ}_{(CH_3COO^-)}}\] = 40.95 S cm2 mol−1)
Chapter:
Give balanced chemical equations involved in the extraction of pure silver.
Chapter:
How will you prepare 1-bromopropane from propene?
Chapter:
How is the following conversion carried out?
Chlorobenzene to aniline.
Chapter:
Explain what happens when a beam of light is passed through a colloidal solution.
Chapter:
Explain what is observed when an electric current is passed through a colloidal sol?
Chapter:
Explain what is observed when an electrolyte (AlCl3) is added to a colloidal solution of arsenious sulphide (As2S3).
Chapter:
How will you bring about the following conversion?
Benzoyl chloride to benzaldehyde.
Chapter:
How will you convert the following: (Give a balanced equation)
Methyl chloride to acetic acid.
Chapter:
How do you convert the following:
Acetic acid to Methane
Chapter:
A ketone A (C4H8O) which undergoes Iodoform reaction gives compound B on reduction. B on heating with conc.H2SO4 at 443 K gives a compound C which forms ozonide D. D on hydrolysis with Zn dust gives only E. Identify the compounds A to E. Write the Iodoform reaction with compound A.
Chapter:
A first-order reaction is 50% complete in 30 minutes at 300 K and in 10 minutes at 320 K. Calculate activation energy (Ea) for the reaction. [R = 8.314 J K−1 mol−1]
[Given: log 2 = 0.3010, log 3 = 0.4771, log 4 = 0.6021]
Chapter:
Advertisements
Give reasons for the following statement:
Transition metals and most of their compounds show paramagnetic behaviour.
Chapter:
Why does the density of transition elements increase from Titanium to Copper? (at. no. Ti = 22, Cu = 29)
Chapter:
Explain the following:
K2Cr2O7 acts as a powerful medium oxidising agent in acidic
Chapter:
The elevation in boiling point when 0.30 g of acetic acid is dissolved in 100 g of benzene is 0.0633°C. Calculate the molecular weight of acetic acid from this data. What conclusion can you draw about the molecular state of the solute in the solution?
(Given Kb for benzene = 2.53 K kg mol−1, at. wt. of C = 12, H = 1, O = 16)
Chapter:
Determine the osmotic pressure of a solution prepared by dissolving 0.025 g of K2SO4 in 2 litres of water at 25°C, assuming that K2SO4 is completely dissociated.
(R = 0.0821 Lit-atm K−1 mol−1, mol. wt. of K2SO4 = 174 g mol−1)
Chapter:
An aqueous solution of a non-volatile solute freezes at 272.4 K, while pure water freezes at 273.0 K. Determine the following:
(Given Kf = 1.86 K kg mol−1, Kb = 0.512 K kg mol−1 and vapour pressure of water at 298 K = 23.756 mm of Hg)
- The molality of solution
- Boiling point of solution
- The lowering of vapour pressure of water at 298 K.
Chapter:
A solution containing 1.23 g of calcium nitrate in 10 g of water, boils at 100.975°C at 760 mm of Hg. Calculate the van’t Hoff factor for the salt at this concentration.
(Kb for water = 0.52 K kg mol−1, mol. wt. of calcium nitrate = 164 g mol−1)
Chapter:
Give the IUPAC name of the following complex:
[Cu(NH3)4]SO4
Chapter:
Give the IUPAC name of the following complex:
[Co(en)2Cl2]
Chapter:
Give the IUPAC name of the following complex:
K3[Al(C2O4)3]
Chapter:
With reference to the coordination complex ion [Fe(H2O)6]2+ answer the following: (at. no. of Fe = 26)
- Give the IUPAC name of the complex ion.
- What is the oxidation number of the central metal atom?
- How many unpaired electrons are there in the complex ion?
- State the type of hybridisation of the complex ion.
Chapter:
Write the type of isomerism exhibited by [Co(NH3)5(NO2)]2+ and [Co(NH3)5ONO]2+ pair of complex ion.
Chapter:
Name the type of isomerism exhibited by the following pair of compounds:
[Cr(H2O)5Cl]Cl2.H2O and [Cr(H2O)4Cl2]Cl.2H2O
Chapter:
Name the type of isomerism exhibited by the following pair of compounds:
[Co(NH3)6][Cr(CN)6] and [Co(CN)6][Cr(NH3)6]
Chapter:
Using the valence bond approach, predict the shape, hybridisation and magnetic behaviour of [Ni(CO)4]. (at. no. of Ni = 28)
Chapter:
What is the action of benzene diazonium chloride on phenol in alkaline medium?
Chapter:
Give a balanced chemical equation for the following reaction:
Diethyl ether is treated with phosphorous pentachloride.
Chapter:
Give a balanced chemical equation for the following reaction:
Ethyl alcohol is treated with thionyl chloride.
Chapter:
Give one chemical test to distinguish between the following pair of compounds:
Ethanol and dimethyl ether
Chapter:
Write the chemical test to distinguish between Propan-1-ol and Propan-2-ol.
Chapter:
Write a chemical equation to illustrate the following name reaction:
Williamson’s synthesis
Chapter:
Write the chemical equation for the following:
Esterification Reaction
Chapter:
Write the equation involved in the following reaction:
Reimer-Tiemann reaction
Chapter:
Identify the compounds A and B in the given reaction:
\[\ce{C2H5OH ->[Cu][\Delta] A ->[CH3COCl][anhy. AlCl3] B}\]
Chapter:
Identify the compounds A and B in the given reaction:
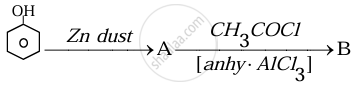
Chapter:
Submit Question Paper
Help us maintain new question papers on Shaalaa.com, so we can continue to help studentsonly jpg, png and pdf files
CISCE previous year question papers ISC Class 12 Chemistry (Theory) with solutions 2019 - 2020
Previous year Question paper for CISCE ISC Class 12 -2020 is solved by experts. Solved question papers gives you the chance to check yourself after your mock test.
By referring the question paper Solutions for Chemistry (Theory), you can scale your preparation level and work on your weak areas. It will also help the candidates in developing the time-management skills. Practice makes perfect, and there is no better way to practice than to attempt previous year question paper solutions of CISCE ISC Class 12.
How CISCE ISC Class 12 Question Paper solutions Help Students ?
• Question paper solutions for Chemistry (Theory) will helps students to prepare for exam.
• Question paper with answer will boost students confidence in exam time and also give you an idea About the important questions and topics to be prepared for the board exam.
• For finding solution of question papers no need to refer so multiple sources like textbook or guides.
