Advertisements
Advertisements
प्रश्न
Write the equation involved in the following reaction:
Reimer-Tiemann reaction
Explain the following with an example.
Reimer-Tiemann reaction
Write a chemical equation to illustrate the following name reaction:
Reimer-Tiemann reaction
Advertisements
उत्तर १
On treating phenol with chloroform in the presence of sodium hydroxide, a -CHO group is introduced at the ortho position of the benzene ring. This reaction is known as the Reimer-Tiemann reaction. The intermediate substituted benzal chloride is hydrolysed in the presence of alkali to produce salicylaldehyde.

उत्तर २
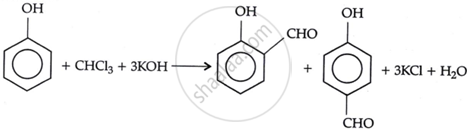
उत्तर ३
Phenol reacts with sodium hydroxide (or KOH) and chloroform in this reaction. 4-hydroxybenzaldehyde is a minor product, while salicylaldehyde is the primary product. Steam distillation can be used to separate it from the p-isomer. Intramolecular hydrogen bonding is responsible for the stability of the p-isomer.

Notes
Students should refer to the answers according to their questions and provide marks.
