Advertisements
Advertisements
प्रश्न
Write the equation involved in the following reaction:
Kolbe’s reaction
Explain the following with an example.
Kolbe’s reaction
रासायनिक समीकरण/संरचनाएँ
स्पष्ट कीजिए
Advertisements
उत्तर
The phenoxide ion produced by the reaction of phenol with sodium hydroxide has greater reactivity than phenol in electrophilic aromatic substitution. Therefore, it experiences electrophilic replacement with carbon dioxide, a mild electrophile. Ortho-hydroxybenzoic acid is produced as the primary reaction product.
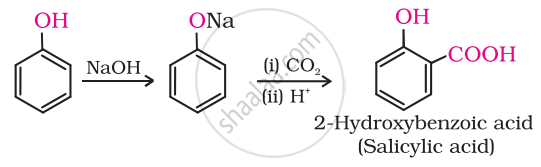
shaalaa.com
Notes
Students should refer to the answer according to their questions.
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
अध्याय 7: Alcohols, Phenols and Ethers - Intext Questions [पृष्ठ २१३]
