Advertisements
Chapters
1: Solutions
2: Electrochemistry
3: Chemical Kinetics
4: The d-block and f-block Elements
5: Coordinate Compounds
Part II
6: Haloalkanes and Haloarenes
▶ 7: Alcohols, Phenols and Ethers
8: Aldehydes, Ketones and Carboxylic Acids
9: Amines
10: Biomolecules
![NCERT solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ chapter 7 - Alcohols, Phenols and Ethers NCERT solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ chapter 7 - Alcohols, Phenols and Ethers - Shaalaa.com](/images/chemistry-part-1-and-2-english-class-12_6:ff01621dcb984083a6bd7b3eaee66bdf.png)
Advertisements
Solutions for Chapter 7: Alcohols, Phenols and Ethers
Below listed, you can find solutions for Chapter 7 of CBSE, Karnataka Board PUC NCERT for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२.
NCERT solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ 7 Alcohols, Phenols and Ethers Intext Questions [Pages 195 - 221]
Classify the following as primary, secondary and tertiary alcohols:
\[\begin{array}{cc}
\ce{CH3}\phantom{.}\\
|\phantom{.....}\\
\ce{CH3 - C - CH2OH}\\
|\phantom{.....}\\
\ce{CH3}\phantom{.}
\end{array}\]
Classify the following as primary, secondary and tertiary alcohols:
H2C = CH – CH2OH
Classify the following as primary, secondary and tertiary alcohols:
CH3 – CH2 – CH2 – OH
Classify the following as primary, secondary and tertiary alcohols:
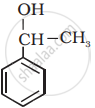
Classify the following as primary, secondary and tertiary alcohols:
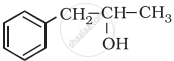
Classify the following as primary, secondary and tertiary alcohols:

Identify allylic alcohols in the following examples.
\[\begin{array}{cc}
\ce{CH3}\phantom{.}\\
|\phantom{.....}\\
\ce{CH3 - C - CH2OH}\\
|\phantom{.....}\\
\ce{CH3}\phantom{.}
\end{array}\]H2C = CH – CH2OH
CH3 – CH2 – CH2 – OH

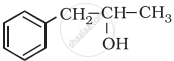

Name the following compound according to the IUPAC system.
\[\begin{array}{cc}
\phantom{..................}\ce{CH2OH}\\
\phantom{.........}|\\
\ce{CH3 - CH2 - CH - CH - CH - CH3}\\
\phantom{.........}|\phantom{...................}|\\
\phantom{.............}\ce{CH2Cl}\phantom{..........}\ce{CH3}\phantom{}
\end{array}\]
Name the following compound according to the IUPAC system.
\[\begin{array}{cc}
\phantom{.....................................}\ce{CH2OH}\\
\phantom{............................}|\\
\ce{CH3 - CH - CH2 - CH - CH - CH3}\\
|\phantom{....................}|\phantom{............}\\
\ce{CH3}\phantom{..............}\ce{OH}\phantom{.........}\\
\end{array}\]
Name the following compound according to the IUPAC system.

Name the following compound according to the IUPAC system.
\[\begin{array}{cc}
\ce{H2C = CH - CH - CH2 - CH2 - CH3}\\
|\phantom{...............}\\
\ce{OH}\phantom{............}
\end{array}\]
Name the following compound according to the IUPAC system.
\[\begin{array}{cc}
\ce{CH3 - C = C - CH2OH}\\
|\phantom{......}|\phantom{.....}\\
\ce{CH3}\phantom{.}\ce{Br}\phantom{....}\\
\end{array}\]
Show how is the following alcohol prepared by the reaction of a suitable Grignard reagent on methanal?
\[\begin{array}{cc}
\ce{CH3 - CH - CH2OH}\\
|\phantom{........}\\
\ce{CH3}\phantom{....}\\
\end{array}\]
Show how is the following alcohol prepared by the reaction of a suitable Grignard reagent on methanal?

\[\ce{CH3 - CH = CH2 ->[H2O/H+]}\]
Write the structure of the product of the following reaction:

Write the structure of the product of the following reaction:
\[\begin{array}{cc}
\ce{CH3 - CH2 - CH - CHO ->[NaBH4]}\\
|\phantom{....}\\
\ce{CH3}\\
\end{array}\]
Give the structure of the product you would expect when the following alcohol reacts with HCl–ZnCl2.
Butan-1-ol
Give the structure of the product you would expect when the following alcohol reacts with HCl–ZnCl2.
2-Methylbutan-2-ol
Give the structure of the product you would expect when the following alcohol reacts with HBr.
Butan-1-ol
Give the structure of the product you would expect when the following alcohol reacts with HBr.
2-Methylbutan-2-ol
Give the structure of the product you would expect when the following alcohol reacts with SOCl2.
Butan-1-ol
Give the structure of the product you would expect when the following alcohol reacts with SOCl2.
2-Methylbutan-2-ol
Predict the major product of acid catalysed dehydration of 1-methylcyclohexanol.
Predict the major product of acid catalysed dehydration of butan-1-ol.
Ortho and para nitrophenols are more acidic than phenol. Draw the resonance structures of the corresponding phenoxide ions.
Write the equation involved in the following reaction:
Reimer-Tiemann reaction
Write the equation involved in the following reaction:
Kolbe’s reaction
Write the reactions of Williamson synthesis of 2-ethoxy-3-methylpentane starting from ethanol and 3-methylpentan-2-ol.
Which of the following is an appropriate set of reactants for the preparation of 1-methoxy-4-nitrobenzene and why?
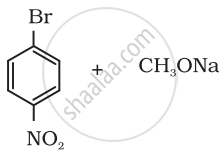 |
 |
| (i) | (ii) |
Predict the product of the following reaction:
\[\ce{CH3 - CH2 - CH2 - O - CH3 + HBr ->}\]
Predict the product of the following reaction:
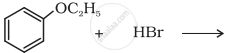
Predict the product of the following reaction:

Predict the product of the following reaction:
\[\ce{(CH3)3C - OC2H5 ->[HI]}\]
NCERT solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ 7 Alcohols, Phenols and Ethers Exercises [Pages 222 - 224]
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{........................}\ce{CH3}\\
\phantom{....................}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{.........}|\phantom{.........}|\\
\phantom{...}\ce{CH3}\phantom{....}\ce{OH}\phantom{....}\ce{CH3}\\
\end{array}\]
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{H3C - CH - CH2 - CH - CH - CH2 - CH3}\\
|\phantom{....................}|\phantom{.........}|\phantom{.............}\\
\ce{OH}\phantom{................}\ce{OH}\phantom{...}\ce{C{_2}H5}\phantom{.........}\\
\end{array}\]
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{CH3 - CH - CH - CH3}\\
|\phantom{.........}|\phantom{...}\\
\ce{OH}\phantom{....}\ce{OH}\\
\end{array}\]
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{HO - CH2 - CH - CH2 - OH}\\
|\phantom{...}\\
\ce{OH}\phantom{.}\\
\end{array}\]
Write the IUPAC name of the following compound:

Write IUPAC name of the following compound:

Write the IUPAC name of the following compound:
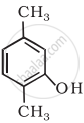
Write the IUPAC name of the following compound:
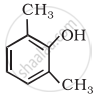
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{CH3 - O - CH2 - CH - CH3}\\
\phantom{................}|\\
\phantom{....................}\ce{CH3}
\end{array}\]
Write the IUPAC name of the following compound:
C6H5 – O – C2H5
Write the IUPAC name of the following compound:
C6H5 – O – C7H15(n−)
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{CH3 - CH2 - O - CH - CH2 - CH3}\\
\phantom{....}|\\
\phantom{........}\ce{CH3}
\end{array}\]
Write the structure of the compound whose IUPAC name is as follows:
2-Methylbutan-2-ol
Write the structure of the compound whose IUPAC name is as follows:
1-Phenylpropan-2-ol
Write the structure of the compound whose IUPAC name is as follows:
3,5-Dimethylhexane-1, 3, 5-triol
Write the structure of the compound whose IUPAC name is as follows:
2,3-Diethylphenol
Write the structure of the compound whose IUPAC name is as follows:
1-Ethoxypropane
Write the structure of the compound whose IUPAC name is as follows:
2-Ethoxy-3-methylpentane
Write the structure of the compound whose IUPAC name is as follows:
Cyclohexylmethanol
Write the structure of the compound whose IUPAC name is as follows:
3-Cyclohexylpentan-3-ol
Write the structure of the compound whose IUPAC name is as follows:
Cyclopent-3-en-1-ol
Write the structure of the compound whose IUPAC name is as follows:
4-Chloro-3-ethylbutan-1-ol
- Draw the structures of all isomeric alcohols of molecular formula C5H12O and give their IUPAC names.
- Classify the isomers of alcohols in the above question as primary, secondary and tertiary alcohols.
Explain why propanol has higher boiling point than that of the hydrocarbon, butane?
Alcohols are comparatively more soluble in water than hydrocarbons of comparable molecular masses. Explain this fact.
What is meant by hydroboration-oxidation reaction? Illustrate it with an example.
Give the structures and IUPAC names of monohydric phenols of molecular formula C7H8O.
While separating a mixture of ortho and para nitrophenols by steam distillation, name the isomer which will be steam volatile. Give reason.
Give the equations of reactions for the preparation of phenol from cumene.
Write chemical reaction for the preparation of phenol from chlorobenzene.
Write the mechanism of hydration of ethene to yield ethanol.
You are given benzene, conc. H2SO4 and NaOH. Write the equations for the preparation of phenol using these reagents.
Show how will you synthesize 1-phenylethanol from a suitable alkene.
Show how will you synthesize cyclohexylmethanol using an alkyl halide by an SN2 reaction.
Show how will you synthesize pentan-1-ol using a suitable alkyl halide.
Give two reactions that show the acidic nature of phenol.
Compare acidity of phenol with that of ethanol.
Give reasons for the following:
o-nitrophenol is more acidic than o-methoxyphenol.
Explain how does the −OH group attached to a carbon of benzene ring activate it towards electrophilic substitution?
Give the equation of the following reaction:
Oxidation of propan-1-ol with alkaline KMnO4 solution.
Give the equation of the following reaction:
Bromine in CS2 with phenol.
Give the equation of the following reaction:
Dilute HNO3 with phenol.
Give the equation of the following reaction:
Treating phenol with chloroform in the presence of aqueous NaOH.
Write the equation involved in the following reaction:
Kolbe’s reaction
Write the equation involved in the following reaction:
Reimer-Tiemann reaction
Explain the following with an example.
Williamson ether synthesis
Explain the following with an example.
Unsymmetrical ether
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
How is the following conversion carried out?
\[\ce{Propene -> Propan-2-ol}\]
How is the following conversion carried out?
\[\ce{Benzyl chloride -> Benzyl alcohol}\]
How is the following conversion carried out?
\[\ce{Ethyl magnesium chloride -> Propan-1-ol}\]
How is the following conversion carried out?
\[\ce{Methyl magnesium bromide -> 2-Methylpropan-2-ol}\]
Name the reagent used in the following reaction:
Oxidation of a primary alcohol to carboxylic acid.
Name the reagent used in the following reaction:
Oxidation of a primary alcohol to aldehyde.
Name the reagent used in the following reaction:
Bromination of phenol to 2,4,6-tribromophenol.
Name the reagent used in the following reaction:
Benzyl alcohol to benzoic acid.
Name the reagent used in the following reaction:
Dehydration of propan-2-ol to propene.
Name the reagent used in the following reaction:
Butan-2-one to butan-2-ol.
Give reason for the higher boiling point of ethanol in comparison to methoxymethane.
Give the IUPAC name of the following ether:
\[\begin{array}{cc}
\ce{C2H5OCH2 - CH - CH3}\\
\phantom{.........}|\\
\phantom{.............}\ce{CH3}
\end{array}\]
Give the IUPAC name of the following ether:
CH3OCH2CH2Cl
Give the IUPAC name of the following ether:
O2N – C6H4 – OCH3(p)
Give the IUPAC name of the following ether:
CH3CH2CH2OCH3
Give the IUPAC name of the following ether:

Give the IUPAC name of the following ether:
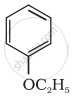
Write the name of the reagent and the equation for the preparation of the following ether by Williamson’s synthesis:
1-Propoxypropane
Write the name of the reagent and the equation for the preparation of the following ether by Williamson’s synthesis:
Ethoxybenzene
Write the name of the reagent and the equation for the preparation of the following ether by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Write the name of the reagent and equation for the preparation of the following ethers by Williamson’s synthesis:
1-Methoxyethane
Illustrate with examples the limitations of Williamson synthesis for the preparation of certain types of ethers.
How is 1-propoxypropane synthesised from propan-1-ol? Write mechanism of this reaction.
Preparation of ethers by acid dehydration of secondary or tertiary alcohols is not a suitable method. Give reason.
Write the equation of the reaction of hydrogen iodide with 1-propoxypropane.
Write the equation of the reaction of hydrogen iodide with methoxybenzene.
Write the equation of the reaction of hydrogen iodide with benzyl ethyl ether.
Explain the fact that in aryl alkyl ethers
- the alkoxy group activates the benzene ring towards electrophilic substitution and
- it directs the incoming substituents to ortho and para positions in the benzene ring.
Write the mechanism of the reaction of HI with methoxymethane.
Write the equation of the following reaction:
Friedel-Crafts reaction - alkylation of anisole
Write the equation of the following reaction:
Nitration of anisole.
Write the equation of the following reaction:
Bromination of anisole in ethanoic acid medium.
Write the equation of the following reaction:
Friedel-Craft’s acetylation of anisole.

Show how you would synthesise the following alcohol from an appropriate alkene?

Show how you would synthesise the following alcohol from an appropriate alkene?
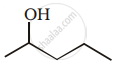
Show how you would synthesise the following alcohol from an appropriate alkene?
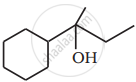
When 3-methylbutan-2-ol is treated with HBr, the following reaction takes place:
\[\begin{array}{cc}
\phantom{...................................}\ce{Br}\\
\phantom{..................................}|\\
\ce{CH3 - CH - CH - CH3 ->[HBr] CH3 - C - CH2 - CH3}\\
|\phantom{.........}|\phantom{...................................}|\phantom{...........}\\
\ce{CH3}\phantom{...}\ce{OH}\phantom{...............................}\ce{CH3}\phantom{.......}\\
\end{array}\]
Give a mechanism for this reaction.
(Hint: The secondary carbocation formed in step II rearranges to a more
stable tertiary carbocation by a hydride ion shift from 3rd carbon atom.)
Solutions for 7: Alcohols, Phenols and Ethers
![NCERT solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ chapter 7 - Alcohols, Phenols and Ethers NCERT solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ chapter 7 - Alcohols, Phenols and Ethers - Shaalaa.com](/images/chemistry-part-1-and-2-english-class-12_6:ff01621dcb984083a6bd7b3eaee66bdf.png)
NCERT solutions for केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ chapter 7 - Alcohols, Phenols and Ethers
Shaalaa.com has the CBSE, Karnataka Board PUC Mathematics केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ CBSE, Karnataka Board PUC solutions in a manner that help students grasp basic concepts better and faster. The detailed, step-by-step solutions will help you understand the concepts better and clarify any confusion. NCERT solutions for Mathematics केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ CBSE, Karnataka Board PUC 7 (Alcohols, Phenols and Ethers) include all questions with answers and detailed explanations. This will clear students' doubts about questions and improve their application skills while preparing for board exams.
Further, we at Shaalaa.com provide such solutions so students can prepare for written exams. NCERT textbook solutions can be a core help for self-study and provide excellent self-help guidance for students.
Concepts covered in केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ chapter 7 Alcohols, Phenols and Ethers are Nomenclature of Alcohols, Phenols and Ethers, Commercially Importance Alcohols, Structures of Functional Groups of Alcohols, Phenols and Ethers, Methods of Preparation of Alcohols, Methods of Preparation of Phenols, Reactions Involving Cleavage of O-H Bond, Overview of Alcohols, Phenols and Ethers, Concept of Alcohols, Phenols and Ethers, Classification of Alcohols, Phenols and Ethers, Physical Properties of Alcohols and Phenols, Chemical Reactions of Alcohols and Phenols, Reactions of Phenols, Reactions Involving Cleavage of Carbon–Oxygen (C–O) Bond in Alcohols, Preparation of Ethers, Physical Properties of Ethers, Chemical Reaction of Ethers - Cleavege of C-O Bonds, Chemical Reaction of Ethers - Electrophilic Substitution, Nomenclature of Alcohols, Phenols and Ethers, Commercially Importance Alcohols, Structures of Functional Groups of Alcohols, Phenols and Ethers, Methods of Preparation of Alcohols, Methods of Preparation of Phenols, Reactions Involving Cleavage of O-H Bond, Overview of Alcohols, Phenols and Ethers, Concept of Alcohols, Phenols and Ethers, Classification of Alcohols, Phenols and Ethers, Physical Properties of Alcohols and Phenols, Chemical Reactions of Alcohols and Phenols, Reactions of Phenols, Reactions Involving Cleavage of Carbon–Oxygen (C–O) Bond in Alcohols, Preparation of Ethers, Physical Properties of Ethers, Chemical Reaction of Ethers - Cleavege of C-O Bonds, Chemical Reaction of Ethers - Electrophilic Substitution.
Using NCERT केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ solutions Alcohols, Phenols and Ethers exercise by students is an easy way to prepare for the exams, as they involve solutions arranged chapter-wise and also page-wise. The questions involved in NCERT Solutions are essential questions that can be asked in the final exam. Maximum CBSE, Karnataka Board PUC केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ students prefer NCERT Textbook Solutions to score more in exams.
Get the free view of Chapter 7, Alcohols, Phenols and Ethers केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ additional questions for Mathematics केमिस्ट्री पार्ट १ एण्ड २ [अंग्रेजी] कक्षा १२ CBSE, Karnataka Board PUC, and you can use Shaalaa.com to keep it handy for your exam preparation.
