Advertisements
Advertisements
प्रश्न
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
Advertisements
उत्तर
In the presence of conc. acids such as H3PO4 and H2SO4 alcohols undergo dehydration to yield alkenes.
\[\ce{\underset{Ethanol}{H3C - CH2 - OH} ->[conc{.} H+/Delta] \underset{Ethene}{CH2 = CH2} + H2O}\]
The mechanism for this reaction is:
Protonation of ethanol (Step 1):
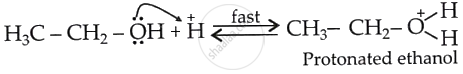
Formation of carbocation (Step 2):
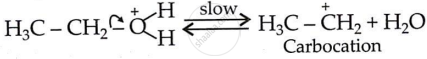
Formation of ethene and regeneration of proton (Step 3):
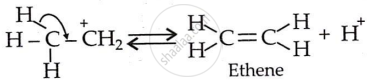
APPEARS IN
संबंधित प्रश्न
Write the mechanism of the following reaction :

Give the equation of the following reaction:
Treating phenol with chloroform in the presence of aqueous NaOH.
When 3-methylbutan-2-ol is treated with HBr, the following reaction takes place:
\[\begin{array}{cc}
\phantom{...................................}\ce{Br}\\
\phantom{..................................}|\\
\ce{CH3 - CH - CH - CH3 ->[HBr] CH3 - C - CH2 - CH3}\\
|\phantom{.........}|\phantom{...................................}|\phantom{...........}\\
\ce{CH3}\phantom{...}\ce{OH}\phantom{...............................}\ce{CH3}\phantom{.......}\\
\end{array}\]
Give a mechanism for this reaction.
(Hint: The secondary carbocation formed in step II rearranges to a more
stable tertiary carbocation by a hydride ion shift from 3rd carbon atom.)
Lucas reagent is ____________.
Which of the following are used to convert RCHO into RCH2OH?
(i) H2/Pd
(ii) LiAlH4
(iii) NaBH4
(iv) Reaction with RMgX followed by hydrolysis
Lucas test is done to differentiate between ____________.
Primary and secondary alcohols on the action of reduced copper give:
During dehydration of alcohols to alkenes by heating with cone. H2SO4 the initial step is ____________.
\[\ce{CH3CH2OH}\] can be converted into \[\ce{CH3CHO}\] by ______.
Identify the secondary alcohols from the following set:
- \[\ce{CH3CH2CH(OH)CH3}\]
- \[\ce{(C2H5)3COH}\]
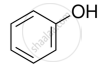
Explain why is OH group in phenols more strongly held as compared to OH group in alcohols.
Explain why nucleophilic substitution reactions are not very common in phenols.
Ethers can be prepared by Williamson synthesis in which an alkyl halide is reacted with sodium alkoxide. Di-tert-butyl ether can’t be prepared by this method. Explain.
What is Lucas reagent?
Which of the following observation is shown by 2-phenyl ethanol with Lucas Reagent?
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
