Advertisements
Advertisements
प्रश्न
Mark the correct increasing order of reactivity of the following compounds with HBr/HCl.
| (a) |  |
| (b) |  |
| (c) |  |
विकल्प
a < b < c
b < a < c
b < c < a
c < b < a
Advertisements
उत्तर
b < c < a
Explanation:
It is type of nucleophilic substitution reaction followed by SN1 mechanism. SN1 mechanism depends on the stability of carbonation. Presence of electron-withdrawing group will decrease the stability of carbocation.
APPEARS IN
संबंधित प्रश्न
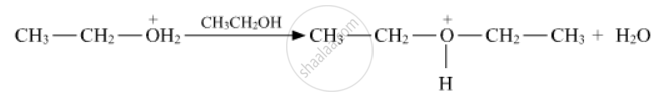
Write the mechanism of the following reaction:

Write the mechanism of the following reaction :

Ortho and para nitrophenols are more acidic than phenol. Draw the resonance structures of the corresponding phenoxide ions.
Show how will you synthesize pentan-1-ol using a suitable alkyl halide.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
Name the reagent used in the following reaction:
Benzyl alcohol to benzoic acid.
Write the mechanism (using curved arrow notation) of the following reaction :
Lucas test is done to differentiate between ____________.
Which one of the following on oxidation gives a ketone?
Cyclohexene is best prepared from cyclohexanol by which of the following:
Name the factors responsible for the solubility of alcohols in water.
The correct geometry around oxygen in CH3OCH3 is
What happens when (CH3)3 C – OH is heated with Cu/573 K?
Write the chemical equation in support of your answer.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
