Advertisements
Advertisements
प्रश्न
Write the mechanism of the following reaction:

Advertisements
उत्तर
The mechanism of the given reaction is as follows:
(i) Protonation of an alcohol molecule
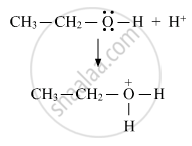
(ii) Attack of other alcohol molecule on the protonated molecule
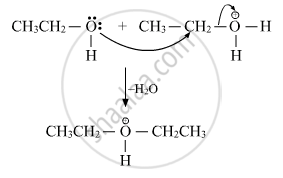
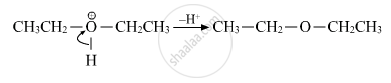
(iii) Deprotonation
APPEARS IN
संबंधित प्रश्न
Ortho and para nitrophenols are more acidic than phenol. Draw the resonance structures of the corresponding phenoxide ions.
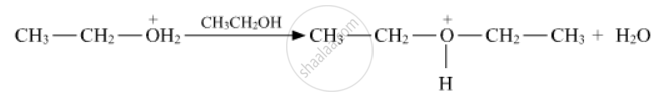
Write the mechanism (using curved arrow notation) of the following reaction :
Lucas reagent is ____________.
By which of the following methods alcohol can be prepared in excellent yield?
Dehydration of 2-butanol yields:
Which one of the following on oxidation gives a ketone?
During dehydration of alcohols to alkenes by heating with cone. H2SO4 the initial step is ____________.
Why is the C – O – H bond angle in alcohols slightly less than the tetrahedral angle whereas the C – O – C bond angle in ether is slightly greater?
What happens when (CH3)3 C – OH is heated with Cu/573 K?
Write the chemical equation in support of your answer.
Write the mechanism of acid dehydration of ethanol to yield ethene.
