Advertisements
Advertisements
प्रश्न
Ortho and para nitrophenols are more acidic than phenol. Draw the resonance structures of the corresponding phenoxide ions.
Advertisements
उत्तर

Resonance structures of o-nitrophenoxide ion

Resonance structures of p-nitrophenoxide ion
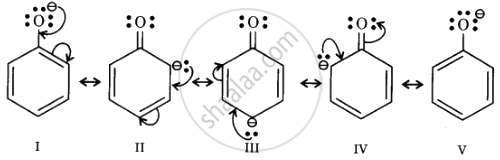
Resonance structures of phenoxide ion
In substituted phenols, electron-withdrawing groups such as the nitro group increase the acidic strength of phenol. This effect becomes more potent when such groups are present at the ortho and para positions. This is because of the effective delocalisation of the anion of the phenoxide ion. Hence, o- and p-nitrophenols are more acidic than phenol.
APPEARS IN
संबंधित प्रश्न
Show how will you synthesize pentan-1-ol using a suitable alkyl halide.
Give the equation of the following reaction:
Treating phenol with chloroform in the presence of aqueous NaOH.
Lucas reagent is ____________.
Which of the following are used to convert RCHO into RCH2OH?
(i) H2/Pd
(ii) LiAlH4
(iii) NaBH4
(iv) Reaction with RMgX followed by hydrolysis
Dehydration of 2-butanol yields:
Which of the following is not true in case of reaction with heated copper at 300°C?
Which one of the following on oxidation gives a ketone?
Primary and secondary alcohols on the action of reduced copper give:
The compound which gives the most stable carbonium ion on dehydration is:
The process of converting alkyl halides into alcohols involves ______.
Name the factors responsible for the solubility of alcohols in water.
Suggest a reagent for the following conversion.

In Kolbe’s reaction, instead of phenol, phenoxide ion is treated with carbon dioxide. Why?
Ethers can be prepared by Williamson synthesis in which an alkyl halide is reacted with sodium alkoxide. Di-tert-butyl ether can’t be prepared by this method. Explain.
The correct geometry around oxygen in CH3OCH3 is
Which of the following observation is shown by 2-phenyl ethanol with Lucas Reagent?
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene
