Advertisements
Advertisements
Question
Ortho and para nitrophenols are more acidic than phenol. Draw the resonance structures of the corresponding phenoxide ions.
Advertisements
Solution

Resonance structures of o-nitrophenoxide ion

Resonance structures of p-nitrophenoxide ion
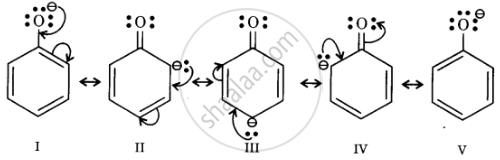
Resonance structures of phenoxide ion
In substituted phenols, electron withdrawing groups such as nitro group increase the acidic strength of phenol. This effect becomes more potent when such groups are present at ortho and para positions. This is because of the effective delocalisation of the anion of the phenoxide ion. Hence, o- and p-nitrophenols are more acidic than phenol.
APPEARS IN
RELATED QUESTIONS
Give reasons for the following:
o-nitrophenol is more acidic than o-methoxyphenol.
Name the reagent used in the following reaction:
Oxidation of a primary alcohol to carboxylic acid.
Name the reagent used in the following reaction:
Benzyl alcohol to benzoic acid.
When 3-methylbutan-2-ol is treated with HBr, the following reaction takes place:
\[\begin{array}{cc}
\phantom{.......................}\ce{Br}\\
\phantom{......................}|\\
\ce{CH3 - CH - CH - CH3 ->[HBr] CH3 - C - CH2 - CH3}\\
\phantom{.}|\phantom{......}|\phantom{......................}|\phantom{........}\\
\phantom{}\ce{CH3}\phantom{...}\ce{OH}\phantom{...................}\ce{CH3}\phantom{.....}
\end{array}\]
Give a mechanism for this reaction.
(Hint: The secondary carbocation formed in step II rearranges to a more
stable tertiary carbocation by a hydride ion shift from 3rd carbon atom.)
By which of the following methods alcohol can be prepared in excellent yield?
Dehydration of 2-butanol yields:
Which of the following is not true in case of reaction with heated copper at 300°C?
Lucas test is done to differentiate between ____________.
\[\ce{CH3CH2OH}\] can be converted into \[\ce{CH3CHO}\] by ______.
Mark the correct increasing order of reactivity of the following compounds with HBr/HCl.
| (a) |  |
| (b) |  |
| (c) |  |
Suggest a reagent for the following conversion.

Explain why nucleophilic substitution reactions are not very common in phenols.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene
