Advertisements
Advertisements
प्रश्न
Ortho and para nitrophenols are more acidic than phenol. Draw the resonance structures of the corresponding phenoxide ions.
Advertisements
उत्तर

Resonance structures of o-nitrophenoxide ion

Resonance structures of p-nitrophenoxide ion
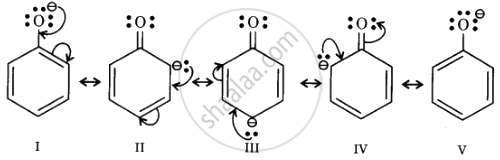
Resonance structures of phenoxide ion
In substituted phenols, electron-withdrawing groups such as the nitro group increase the acidic strength of phenol. This effect becomes more potent when such groups are present at the ortho and para positions. This is because of the effective delocalisation of the anion of the phenoxide ion. Hence, o- and p-nitrophenols are more acidic than phenol.
APPEARS IN
संबंधित प्रश्न
Give reasons for the following:
o-nitrophenol is more acidic than o-methoxyphenol.
Write the mechanism of the following reaction :

Give the equation of the following reaction:
Treating phenol with chloroform in the presence of aqueous NaOH.
Name the reagent used in the following reaction:
Oxidation of a primary alcohol to carboxylic acid.
Lucas reagent is ____________.
The compound which reacts fastest with Lucas reagent at room temperature is:
By which of the following methods alcohol can be prepared in excellent yield?
Dehydration of 2-butanol yields:
Suggest a reagent for the following conversion.

Ethers can be prepared by Williamson synthesis in which an alkyl halide is reacted with sodium alkoxide. Di-tert-butyl ether can’t be prepared by this method. Explain.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene
