Advertisements
Advertisements
प्रश्न
The process of converting alkyl halides into alcohols involves ______.
पर्याय
addition reaction
substitution reaction
dehydrohalogenation reaction
rearrangement reaction
Advertisements
उत्तर
The process of converting alkyl halides into alcohols involves substitution reaction.
Explanation:
Conversion of alkyl halides into alcohols involves substitution reaction.
\[\ce{RX ->[NaOH] ROH + NaX}\]
APPEARS IN
संबंधित प्रश्न
Give the equation of the following reaction:
Oxidation of propan-1-ol with alkaline KMnO4 solution.
Give the equation of the following reaction:
Treating phenol with chloroform in the presence of aqueous NaOH.
Name the reagent used in the following reaction:
Oxidation of a primary alcohol to carboxylic acid.
Name the reagent used in the following reaction:
Benzyl alcohol to benzoic acid.
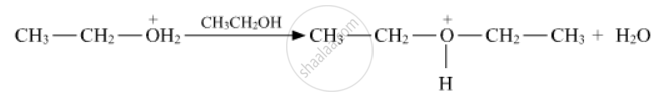
Write the mechanism (using curved arrow notation) of the following reaction :
Lucas test is used for the detection of _____________.
By which of the following methods alcohol can be prepared in excellent yield?
Which of the following is not true in case of reaction with heated copper at 300°C?
Primary and secondary alcohols on the action of reduced copper give:
Cyclohexene is best prepared from cyclohexanol by which of the following:
During dehydration of alcohols to alkenes by heating with cone. H2SO4 the initial step is ____________.
Mark the correct increasing order of reactivity of the following compounds with HBr/HCl.
| (a) |  |
| (b) |  |
| (c) |  |
Name the factors responsible for the solubility of alcohols in water.
Explain why is OH group in phenols more strongly held as compared to OH group in alcohols.
In Kolbe’s reaction, instead of phenol, phenoxide ion is treated with carbon dioxide. Why?
What happens when (CH3)3 C – OH is heated with Cu/573 K?
Write the chemical equation in support of your answer.
Which of the following alcohols will not undergo oxidation?
Write the mechanism of acid dehydration of ethanol to yield ethene.
