Advertisements
Advertisements
प्रश्न
Mark the correct increasing order of reactivity of the following compounds with HBr/HCl.
| (a) |  |
| (b) |  |
| (c) |  |
पर्याय
a < b < c
b < a < c
b < c < a
c < b < a
Advertisements
उत्तर
b < c < a
Explanation:
It is type of nucleophilic substitution reaction followed by SN1 mechanism. SN1 mechanism depends on the stability of carbonation. Presence of electron-withdrawing group will decrease the stability of carbocation.
APPEARS IN
संबंधित प्रश्न
Write the final product(s) in each of the following reactions:

Ortho and para nitrophenols are more acidic than phenol. Draw the resonance structures of the corresponding phenoxide ions.
Give the equation of the following reaction:
Oxidation of propan-1-ol with alkaline KMnO4 solution.
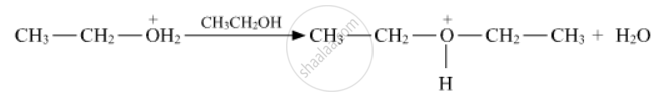
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
Write the mechanism (using curved arrow notation) of the following reaction :
Lucas test is done to differentiate between ____________.
Which one of the following on oxidation gives a ketone?
Cyclohexene is best prepared from cyclohexanol by which of the following:
During dehydration of alcohols to alkenes by heating with cone. H2SO4 the initial step is ____________.
Identify the secondary alcohols from the following set:
- \[\ce{CH3CH2CH(OH)CH3}\]
- \[\ce{(C2H5)3COH}\]
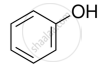
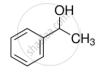
The process of converting alkyl halides into alcohols involves ______.
Suggest a reagent for the following conversion.

In Kolbe’s reaction, instead of phenol, phenoxide ion is treated with carbon dioxide. Why?
What is Lucas reagent?
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene
