Advertisements
Advertisements
प्रश्न
\[\ce{CH3CH2OH}\] can be converted into \[\ce{CH3CHO}\] by ______.
पर्याय
catalytic hydrogenation
treatment with \[\ce{LiAlH4}\]
treatment with pyridinium chlorochromate
treatment with \[\ce{KMnO4}\]
Advertisements
उत्तर
\[\ce{CH3CH2OH}\] can be converted into \[\ce{CH3CHO}\] by treatment with pyridinium chlorochromate.
Explanation:
Alcohols are oxidized to aldehydes and finally to acids.
\[\ce{CH3CH2OH ->[{[O]}] CH3CHO ->[{[O]}] CH3COOH}\]
Oxidations can be stooped at aldehyde stage by using pyridinium chlorochromate
\[\ce{(CrO3C5H5N.HCl)}\].
\[\ce{CH3CH2OH ->[PCC] CH3CHO}\]
APPEARS IN
संबंधित प्रश्न
Ortho and para nitrophenols are more acidic than phenol. Draw the resonance structures of the corresponding phenoxide ions.
Show how will you synthesize pentan-1-ol using a suitable alkyl halide.
Give the equation of the following reaction:
Oxidation of propan-1-ol with alkaline KMnO4 solution.
Give the equation of the following reaction:
Dilute HNO3 with phenol.
Give the equation of the following reaction:
Treating phenol with chloroform in the presence of aqueous NaOH.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
Name the reagent used in the following reaction:
Oxidation of a primary alcohol to aldehyde.
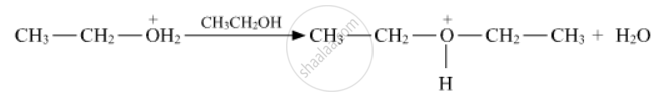
Write the mechanism (using curved arrow notation) of the following reaction :
Which of the following are used to convert RCHO into RCH2OH?
(i) H2/Pd
(ii) LiAlH4
(iii) NaBH4
(iv) Reaction with RMgX followed by hydrolysis
Which of the following is not true in case of reaction with heated copper at 300°C?
Primary and secondary alcohols on the action of reduced copper give:
Cyclohexene is best prepared from cyclohexanol by which of the following:
Alcohols react with active metals e.g. Na, K etc. to give corresponding alkoxides. Write down the decreasing order of reactivity of sodium metal towards primary, secondary and tertiary alcohols.
Ethers can be prepared by Williamson synthesis in which an alkyl halide is reacted with sodium alkoxide. Di-tert-butyl ether can’t be prepared by this method. Explain.
The correct geometry around oxygen in CH3OCH3 is
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene
