Advertisements
Advertisements
प्रश्न
Alcohols react with active metals e.g. Na, K etc. to give corresponding alkoxides. Write down the decreasing order of reactivity of sodium metal towards primary, secondary and tertiary alcohols.
Advertisements
उत्तर
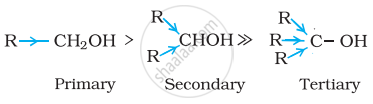
Decreasing order of reactivity of sodium metal is:
1° > 2° > 3°
Alcohols react with sodium metal to form alkoxides and hydrogen is liberated:
\[\ce{R - O - H + Na -> RO - Na+ {+} 1/2 H2}\]
The order of reactivity of alcohols is primary > secondary > tertiary. This can be explained on the basis of cleavage of O – H bond. The alkyl groups are electron releasing groups (+I effect) and they increase the electron density around the oxygen. As a result, the electrons of O – H bond cannot be withdrawn strongly towards oxygen and O – H remains strong. Therefore, greater is the number, of alkyl groups present, smaller will be reactivity of alcohol.
APPEARS IN
संबंधित प्रश्न
Write the mechanism of the following reaction :

Give the equation of the following reaction:
Oxidation of propan-1-ol with alkaline KMnO4 solution.
Give the equation of the following reaction:
Dilute HNO3 with phenol.
The compound which reacts fastest with Lucas reagent at room temperature is:
In the reduction \[\ce{R - CHO + H2 -> RCH2OH}\] the catalyst used is:
Which of the following is not true in case of reaction with heated copper at 300°C?
Primary and secondary alcohols on the action of reduced copper give:
The compound which gives the most stable carbonium ion on dehydration is:
Identify the secondary alcohols from the following set:
- \[\ce{CH3CH2CH(OH)CH3}\]
- \[\ce{(C2H5)3COH}\]
Name the factors responsible for the solubility of alcohols in water.
Why is the C – O – H bond angle in alcohols slightly less than the tetrahedral angle whereas the C – O – C bond angle in ether is slightly greater?
Assertion: Bond angle in ethers is slightly less than the tetrahedral angle.
Reason: There is a repulsion between the two bulky (–R) groups.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
