Advertisements
Advertisements
प्रश्न
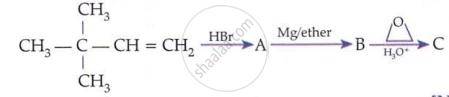
Arrange the following compounds in increasing order of acidity and give a suitable explanation.
Phenol, o-nitrophenol, o-cresol
Advertisements
उत्तर
Increasing order of acidity.
o-cresol < Phenol < o-nitrophenol
In cresol, 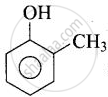 , the electron-donating group \[\ce{(-CH3)}\] gives electrons and intensify the charge on phenoxide ion and therefore makes it unstable. Therefore, o-cresol is less acidic than phenol. In o-nitrophenol, the electron-withdrawing \[\ce{(-NO2)}\] group withdraws electrons and disperses the - ve charge and stabilizes the phenoxide ion. Therefore, o-nitrophenol is more acidic than phenol.
, the electron-donating group \[\ce{(-CH3)}\] gives electrons and intensify the charge on phenoxide ion and therefore makes it unstable. Therefore, o-cresol is less acidic than phenol. In o-nitrophenol, the electron-withdrawing \[\ce{(-NO2)}\] group withdraws electrons and disperses the - ve charge and stabilizes the phenoxide ion. Therefore, o-nitrophenol is more acidic than phenol.
APPEARS IN
संबंधित प्रश्न
Write resonance structures of aniline. What is the action of benzene diazonium chloride on ethanol?
Give simple chemical test to distinguish between Ethanol and Phenol.
Which of the following is correct?
Wood spirit is known as acetone:
Out of 2-chloroethanol and ethanol which is more acidic and why?
Name the enzymes and write the reactions involved in the preparation of ethanol from sucrose by fermentation.
Dipole moment of phenol is smaller than that of methanol. Why?
Match the items of column I with items of column II.
| Column I | Column II | |
| (i) | Antifreeze used in car engine | (a) Neutral ferric chloride |
| (ii) | Solvent used in perfumes | (b) Glycerol |
| (iii) | Starting material for picric acid | (c) Methanol |
| (iv) | Wood spirit | (d) Phenol |
| (v) | Reagent used for detection of phenolic group | (e) Ethleneglycol |
| (vi) | By product of soap industry used in cosmetics | (f) Ethanol |
Match the items of column I with items of column II.
| Column I | Column II | |
| (i) | Methanol | (a) Conversion of phenol to o-hydroxysalicylic acid |
| (ii) | Kolbe’s reaction | (b) Ethyl alcohol |
| (iii) | Williamson’s synthesis | (c) Conversion of phenol to salicylaldehyde |
| (iv) | Conversion of 2° alcohol to ketone | (d) Wood spirit |
| (v) | Reimer-Tiemann reaction | (e) Heated copper at 573 K |
| (vi) | Fermentation | (f) Reaction of alkyl halide with sodium alkoxide |
Assertion: Ethanol is a weaker acid than phenol.
Reason: Sodium ethoxide may be prepared by the reaction of ethanol with aqueous \[\ce{NaOH}\].
Convert the following:
Propanenitrile to ethanol.
Liquor poisoning is due to
Convert the following Ethanal to ethanol.
Give IUPAC names of the following compounds:
\[\begin{array}{cc}
\ce{CH3 - CH - CH - CH - CH2-OH }\\
|\phantom{......}|\phantom{......}|\phantom{.......}\\
\ce{Cl}\phantom{....}\ce{CH3}\phantom{...}\ce{CH3}\phantom{.....}
\end{array}\]
3, 3-dimethyl-1-butene was subject to following reactions: