Advertisements
Advertisements
प्रश्न
Arrange water, ethanol and phenol in increasing order of acidity and give reason for your answer.
Advertisements
उत्तर
The increasing order of acidity is ethanol < water < phenol. The phenoxide ion obtained after the removal of a proton is stabilised by resonance whereas the ethoxide ion obtained after the removal of a proton is destabilised by the ‘+I’ effect of the —C2H5 group. Therefore phenol is a stronger acid than ethanol. On the other hand, ethanol is a weaker acid than water because the electron-releasing —C2H5 group in ethanol increases the electron density on oxygen and hence the polarity of the O—H bond in ethanol decreases which results in the decreasing acidic strength. Hence acidic strength increases in the order given above.
APPEARS IN
संबंधित प्रश्न
How do you convert the following:
Ethanol to Propan-2-ol
Write the mechanism of the following reaction :
\[\ce{C2H5OH->[H2SO4][443K]CH2=CH2 + H2O}\]
Among the following the one which reacts most readily with ethanol is:
Which of the following is correct?
Out of 2-chloroethanol and ethanol which is more acidic and why?
Suggest a reagent for conversion of ethanol to ethanal.
Suggest a reagent for conversion of ethanol to ethanoic acid.
Dipole moment of phenol is smaller than that of methanol. Why?
Match the items of column I with items of column II.
| Column I | Column II | |
| (i) | Methanol | (a) Conversion of phenol to o-hydroxysalicylic acid |
| (ii) | Kolbe’s reaction | (b) Ethyl alcohol |
| (iii) | Williamson’s synthesis | (c) Conversion of phenol to salicylaldehyde |
| (iv) | Conversion of 2° alcohol to ketone | (d) Wood spirit |
| (v) | Reimer-Tiemann reaction | (e) Heated copper at 573 K |
| (vi) | Fermentation | (f) Reaction of alkyl halide with sodium alkoxide |
Convert the following:
Propanenitrile to ethanol.
Tonics in general contain
Liquor poisoning is due to
Write chemical reactions of following reagents on methoxyethane:
dilute H2SO4
How methanol is obtained from methanal.
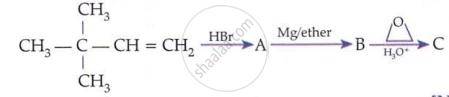
3, 3-dimethyl-1-butene was subject to following reactions: