Advertisements
Advertisements
प्रश्न
Match the structures of the compounds given in Column I with the name of the compounds given in Column II.
| Column I | Column II | |
| (i) |  |
(a) Hydroquinone |
| (ii) |  |
(b) Phenetole |
| (iii) | 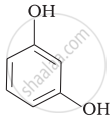 |
(c) Catechol |
| (iv) |  |
(d) o-Cresol |
| (v) |  |
(e) guinone |
| (vi) | 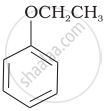 |
(f) Resorcinol |
| (g) Anisole |
Advertisements
उत्तर
| Column I | Column II | |
| (i) |  |
(d) o-Cresol |
| (ii) |  |
(c) Catechol |
| (iii) |  |
(f) Resorcinol |
| (iv) |  |
(a) Hydroquinone |
| (v) |  |
(g) Anisole |
| (vi) |  |
(b) Phenetole |
Explanation:
(i) Cresols are organic compounds which are methyl phenols. There are three forms of cresol: o-cresol, p-cresol and m-cresol.
(ii) Catechol is also known as pyrocatechol. Its IUPAC name is 1, 2-dihydrobenzene. It is used in the production of pesticides, perfumes and pharmaceuticals.
(iii) Its IUPAC name is 1, 3-dihydroxybenzene. Resorcinol is used to treat acne, seborrheic dermatitis and other skin disorder.
(iv) Hydroquinone is also known as quinol. Its IUPAC name is 1, 4-dihydroxybenzene. It is a white granular solid. It is a good reducing agent.
(v) Anisole or methoxy benzene, is a colourless liquid with a smell reminiscent of an anise seed.
(vi) Phenetole is an organic compound. It is also known as ethyl phenyl ether. It is volatile in nature and its vapour is explosive in nature.
APPEARS IN
संबंधित प्रश्न
Explain metamerism with suitable examples of ethers
Name the following compound according to the IUPAC system.

Name the following compound according to the IUPAC system.
\[\begin{array}{cc}
\ce{CH3 - C = C - CH2OH}\\
|\phantom{......}|\phantom{.....}\\
\ce{CH3}\phantom{.}\ce{Br}\phantom{....}\\
\end{array}\]
Write the IUPAC name of the following compound:
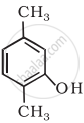
- Draw the structures of all isomeric alcohols of molecular formula C5H12O and give their IUPAC names.
- Classify the isomers of alcohols in the above question as primary, secondary and tertiary alcohols.
Give the IUPAC name of the following ether:
O2N – C6H4 – OCH3(p)
In the dehydration of alcohols to alkenes by heating with concentrated sulphuric acid, the initiation step is:
(1) formation of carbonation
(2) formation of an ester
(3) protonation of the alcohol molecule
(4) elimination of water
Write structural formulae for 1-Ethylcyclohexanol.
An example of a compound with functional group – O – is ____________.
HBr reacts fastest with ____________.
When ethyl alcohol reacts with acetic acid, the products formed are:
The IUPAC name of the ether CH2 = CH–CH2OCH3 is:
\[\ce{HC ≡ CH ->[HgSO4][H2SO4] ->[CH3MgBr][H2O] ->[PBr3]}\]
IUPAC name of the compound is:
\[\begin{array}{cc}
\ce{CH3-CH-OCH3}\\
|\phantom{....}\\
\ce{CH3}\phantom{..}
\end{array}\]
Assertion: IUPAC name of the compound
\[\begin{array}{cc}
\ce{CH3 - CH - O - CH2 - CH2 - CH3}\\
|\phantom{....................}\\
\ce{CH3}\phantom{.................}
\end{array}\] is 2-Ethoxy-2-methylethane.
Reason: In IUPAC nomenclature, ether is regarded as hydrocarbon derivative in which a hydrogen atom is replaced by —OR or —OAr group [where R = alkyl group and Ar = aryl group]
Assertion: Like bromination of benzene, bromination of phenol is also carried out in the presence of Lewis acid.
Reason: Lewis acid polarises the bromine molecule.
Identify A and B in the following:

How are the following conversions carried out?
Methyl magnesium bromide→2-Methylpropan-2-ol.
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{................}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3}\phantom{..}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
Write the IUPAC name.
\[\begin{array}{cc}
\phantom{................}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C -CH3}\\
\phantom{.}|\phantom{......}|\phantom{......}|\\
\phantom{....}\ce{CH3\phantom{...}\ce{OH}\phantom{...}\ce{CH3}}\
\end{array}\]
