Advertisements
Advertisements
Question
Match the structures of the compounds given in Column I with the name of the compounds given in Column II.
| Column I | Column II | |
| (i) |  |
(a) Hydroquinone |
| (ii) |  |
(b) Phenetole |
| (iii) | 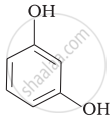 |
(c) Catechol |
| (iv) |  |
(d) o-Cresol |
| (v) |  |
(e) guinone |
| (vi) | 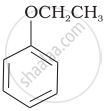 |
(f) Resorcinol |
| (g) Anisole |
Advertisements
Solution
| Column I | Column II | |
| (i) |  |
(d) o-Cresol |
| (ii) |  |
(c) Catechol |
| (iii) |  |
(f) Resorcinol |
| (iv) |  |
(a) Hydroquinone |
| (v) |  |
(g) Anisole |
| (vi) |  |
(b) Phenetole |
Explanation:
(i) Cresols are organic compounds which are methyl phenols. There are three forms of cresol: o-cresol, p-cresol and m-cresol.
(ii) Catechol is also known as pyrocatechol. Its IUPAC name is 1, 2-dihydrobenzene. It is used in the production of pesticides, perfumes and pharmaceuticals.
(iii) Its IUPAC name is 1, 3-dihydroxybenzene. Resorcinol is used to treat acne, seborrheic dermatitis and other skin disorder.
(iv) Hydroquinone is also known as quinol. Its IUPAC name is 1, 4-dihydroxybenzene. It is a white granular solid. It is a good reducing agent.
(v) Anisole or methoxy benzene, is a colourless liquid with a smell reminiscent of an anise seed.
(vi) Phenetole is an organic compound. It is also known as ethyl phenyl ether. It is volatile in nature and its vapour is explosive in nature.
APPEARS IN
RELATED QUESTIONS
Write IUPAC name of the following compound:

Give the IUPAC name of the following ether:
O2N – C6H4 – OCH3(p)
Natalite is a mixture of
(a) diethyl ether and methanol
(b) diethyl ether and ethanol
(c) dimethyl ether and methanol
(d) dimethyl ether and ethanol
How is phenol converted into the following?
Benzene
How is phenol converted into the following?
picric acid
Propanoic acid to ethylamine.
Write IUPAC name of the following
\[\begin{array}{cc}\ce{CH3-CH-CH-CH2-OH}\\|\phantom{.....}|\phantom{.......}\\\ce{OH}\phantom{..}\ce{CH3}\phantom{.....}\end{array}\]
Which of the following compounds is oxidised to prepare methyl ethyl ketone?
1-Propanol and 2-propanol can be best distinguished by:
The major product formed by the reaction:
\[\begin{array}{cc}
\ce{CH3CH-CH2Br ->[CH3O^-][CH3OH] is}\\
|\phantom{................}\\
\ce{CH3}\phantom{.............}
\end{array}\]
Which of the following gives a positive iodoform test?
Give IUPAC name of the compound given below.
\[\begin{array}{cc}
\phantom{}\ce{CH3 - CH - CH2 - CH2 - CH - CH3}\phantom{.}\\
\phantom{.........}|\phantom{...................}|\phantom{...........}\\
\phantom{..}\ce{Cl}\phantom{.................}\ce{OH}\phantom{..}
\end{array}\]
Which of the following reagents can be used to oxidise primary alcohols to aldehydes?
(i) \[\ce{CrO3}\] in anhydrous medium.
(ii) \[\ce{KMnO4}\] in acidic medium.
(iii) Pyridinium chlorochromate.
(iv) Heat in the presence of Cu at 573 K.
What happens when benzene diazonium chloride is heated with water?
Assertion: Addition reaction of water to but-1-ene in acidic medium yields butan-1-ol.
Reason: Addition of water in acidic medium proceeds through the formation of primary carbocation.
Assertion: Phenol forms 2, 4, 6 – tribromophenol on treatment with \[\ce{Br2}\] in carbon disulphide at 273 K.
Reason: Bromine polarises in carbon disulphide.
Draw structure of the following compound.
2. 5-Diethylphenol
Write structural formulae for:
Salicylic acid
Write IUPAC names of the following compounds:
\[\begin{array}{cc}
\phantom{...............}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3\phantom{...}OH\phantom{...}CH3}\\
\end{array}\]
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{................}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3}\phantom{...}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
