Advertisements
Advertisements
Question
Which of the following reagents can be used to oxidise primary alcohols to aldehydes?
(i) \[\ce{CrO3}\] in anhydrous medium.
(ii) \[\ce{KMnO4}\] in acidic medium.
(iii) Pyridinium chlorochromate.
(iv) Heat in the presence of Cu at 573 K.
Advertisements
Solution
(i) \[\ce{CrO3}\] in anhydrous medium.
(iii) Pyridinium chlorochromate.
(iv) Heat in the presence of Cu at 573 K.
Explanation:
Strong oxidising agents such as acidified potassium permanganate are used for getting carboxylic acids from alcohols directly. \[\ce{CrO3}\] in anhydrous medium is used as the oxidising agent for the isolation of aldehydes.
\[\ce{RCH2OH ->[CrO3] RCHO}\]
A better reagent for the oxidation of primary alcohols to aldehydes in good yield is pyridinium chlorochromate (PCC), a complex for chromium trioxide with pyridine and HCl.
APPEARS IN
RELATED QUESTIONS
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{CH3 - O - CH2 - CH - CH3}\\
\phantom{................}|\\
\phantom{....................}\ce{CH3}
\end{array}\]
Give the IUPAC name of the following ether:
\[\begin{array}{cc}
\ce{C2H5OCH2 - CH - CH3}\\
\phantom{.........}|\\
\phantom{.............}\ce{CH3}
\end{array}\]
Give the IUPAC name of the following ether:
CH3OCH2CH2Cl
Write the structure and IUPAC name of 'methyl-n-propyl ether'.
In the dehydration of alcohols to alkenes by heating with concentrated sulphuric acid, the initiation step is:
(1) formation of carbonation
(2) formation of an ester
(3) protonation of the alcohol molecule
(4) elimination of water
Write structural formula for Methyl vinyl ether.
Write structural formulae for Cyclohex-2-en-1-ol.
Give IUPAC names of the following compound:
\[\begin{array}{cc}
\phantom{..}\ce{H}\phantom{...}\ce{CH3}\phantom{.}\ce{H}\phantom{..}\\
\phantom{}|\phantom{....}|\phantom{....}|\phantom{}\\
\ce{H - C - C - C - H}\\
\phantom{}|\phantom{....}|\phantom{....}|\phantom{}\\
\phantom{.}\ce{H}\phantom{...}\ce{OH}\phantom{.}\ce{H}\phantom{.}\\
\end{array}\]
The compound HOCH2 – CH2OH is __________.
An example of a compound with functional group – O – is ____________.
Butane-2-ol is ____________.
HBr reacts fastest with ____________.
Write steps to carry out the conversion of phenol to aspirin.
Match the starting materials given in Column I with the products formed by these (Column II) in the reaction with HI.
| Column I | Column II | ||
| (i) | CH3—O—CH3 | (a) |  |
| (ii) | \[\begin{array}{cc} \ce{CH3}\phantom{..................}\\ \backslash\phantom{.............}\\ \ce{CH-O-CH3}\\ /\phantom{..............}\\ \ce{CH3}\phantom{..................} \end{array}\] |
(b) | \[\begin{array}{cc} \ce{CH3}\phantom{....}\\ |\phantom{.......}\\ \ce{CH3-C-I + CH3OH}\\ |\phantom{.......}\\ \ce{CH3}\phantom{....} \end{array}\] |
| (iii) | \[\begin{array}{cc} \ce{CH3}\phantom{.}\\ |\phantom{....}\\ \ce{H3C-C-O-CH3}\\ |\phantom{....}\\ \ce{CH3}\phantom{..} \end{array}\] |
(c) | 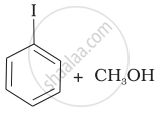 |
| (iv) |  |
(d) | CH3—OH + CH3—I |
| (e) | \[\begin{array}{cc} \ce{CH3}\phantom{.....................}\\ \backslash\phantom{.................}\\ \ce{CH-OH + CH3I}\\ /\phantom{.................}\\ \ce{CH3}\phantom{.....................} \end{array}\] |
||
| (f) | \[\begin{array}{cc} \ce{CH3}\phantom{.....................}\\ \backslash\phantom{.................}\\ \ce{CH-I + CH3OH}\\ /\phantom{.................}\\ \ce{CH3}\phantom{.....................} \end{array}\] |
||
| (g) | \[\begin{array}{cc} \ce{CH3}\phantom{....}\\ |\phantom{.......}\\ \ce{CH3-C-OH + CH3I}\\ |\phantom{.......}\\ \ce{CH3}\phantom{....} \end{array}\] |
Assertion: Like bromination of benzene, bromination of phenol is also carried out in the presence of Lewis acid.
Reason: Lewis acid polarises the bromine molecule.
Explain why Lewis acid is not required in bromination of phenol?
How can phenol be converted to aspirin?
Write the IUPAC name of the following compound.

Write structural formulae for:
Salicylic acid
The IUPAC name of 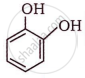 is ______.
is ______.
