Advertisements
Advertisements
प्रश्न
Which of the following reagents can be used to oxidise primary alcohols to aldehydes?
(i) \[\ce{CrO3}\] in anhydrous medium.
(ii) \[\ce{KMnO4}\] in acidic medium.
(iii) Pyridinium chlorochromate.
(iv) Heat in the presence of Cu at 573 K.
Advertisements
उत्तर
(i) \[\ce{CrO3}\] in anhydrous medium.
(iii) Pyridinium chlorochromate.
(iv) Heat in the presence of Cu at 573 K.
Explanation:
Strong oxidising agents such as acidified potassium permanganate are used for getting carboxylic acids from alcohols directly. \[\ce{CrO3}\] in anhydrous medium is used as the oxidising agent for the isolation of aldehydes.
\[\ce{RCH2OH ->[CrO3] RCHO}\]
A better reagent for the oxidation of primary alcohols to aldehydes in good yield is pyridinium chlorochromate (PCC), a complex for chromium trioxide with pyridine and HCl.
APPEARS IN
संबंधित प्रश्न
Name the following compound according to the IUPAC system.
\[\begin{array}{cc}
\phantom{.....................................}\ce{CH2OH}\\
\phantom{............................}|\\
\ce{CH3 - CH - CH2 - CH - CH - CH3}\\
|\phantom{....................}|\phantom{............}\\
\ce{CH3}\phantom{..............}\ce{OH}\phantom{.........}\\
\end{array}\]
Name the following compound according to the IUPAC system.
\[\begin{array}{cc}
\ce{H2C = CH - CH - CH2 - CH2 - CH3}\\
|\phantom{...............}\\
\ce{OH}\phantom{............}
\end{array}\]
Write the IUPAC name of the following compound:
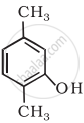
Write structures of the compounds whose IUPAC names are as follows:
3-Chloromethylpentan-1-ol.
- Draw the structures of all isomeric alcohols of molecular formula C5H12O and give their IUPAC names.
- Classify the isomers of alcohols in the above question as primary, secondary and tertiary alcohols.
Give the IUPAC name of the following ether:
CH3CH2CH2OCH3
Ethylidene dichloride when boiled with aqueous solution of NaOH yields _______.
(A) formaldehyde
(B) acetaldehyde
(C) acetone
(D) ethyl methyl ketone
Write IUPAC name of the following compound (CH3)2 N − CH2CH3
Give reasons Fluoride ion has higher hydration enthalpy than chloride ion.
How do you convert the Ethanal to Propanone
Propanoic acid to ethylamine.
Write structural formula for pentane-1,4-diol.
Write IUPAC name of the following
\[\begin{array}{cc}\ce{CH3-CH-CH-CH2-OH}\\|\phantom{.....}|\phantom{.......}\\\ce{OH}\phantom{..}\ce{CH3}\phantom{.....}\end{array}\]
HBr reacts fastest with ____________.
Which of the following gives a positive iodoform test?
Give IUPAC name of the compound given below.
\[\begin{array}{cc}
\phantom{}\ce{CH3 - CH - CH2 - CH2 - CH - CH3}\phantom{.}\\
\phantom{.........}|\phantom{...................}|\phantom{...........}\\
\phantom{..}\ce{Cl}\phantom{.................}\ce{OH}\phantom{..}
\end{array}\]
Write the IUPAC name of the compound given below.
\[\begin{array}{cc}
\phantom{}\ce{CH3 - CH2 - C = C - OH}\\
\phantom{........}|\phantom{....}|\phantom{}\\
\phantom{..............}\ce{CH3 CH2OH}
\end{array}\]
The IUPAC name of 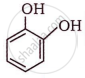 is ______.
is ______.
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{..............}\ce{CH3}\\
\phantom{............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{.....}|\phantom{......}|\\
\phantom{...}\ce{CH3}\phantom{.}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
Write IUPAC names of the following compounds:
\[\begin{array}{cc}
\phantom{...............}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3\phantom{...}OH\phantom{...}CH3}\\
\end{array}\]
