Advertisements
Advertisements
प्रश्न
Which of the following reagents can be used to oxidise primary alcohols to aldehydes?
(i) \[\ce{CrO3}\] in anhydrous medium.
(ii) \[\ce{KMnO4}\] in acidic medium.
(iii) Pyridinium chlorochromate.
(iv) Heat in the presence of Cu at 573 K.
Advertisements
उत्तर
(i) \[\ce{CrO3}\] in anhydrous medium.
(iii) Pyridinium chlorochromate.
(iv) Heat in the presence of Cu at 573 K.
Explanation:
Strong oxidising agents such as acidified potassium permanganate are used for getting carboxylic acids from alcohols directly. \[\ce{CrO3}\] in anhydrous medium is used as the oxidising agent for the isolation of aldehydes.
\[\ce{RCH2OH ->[CrO3] RCHO}\]
A better reagent for the oxidation of primary alcohols to aldehydes in good yield is pyridinium chlorochromate (PCC), a complex for chromium trioxide with pyridine and HCl.
APPEARS IN
संबंधित प्रश्न
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{H3C - CH - CH2 - CH - CH - CH2 - CH3}\\
|\phantom{....................}|\phantom{.........}|\phantom{.............}\\
\ce{OH}\phantom{................}\ce{OH}\phantom{...}\ce{C{_2}H5}\phantom{.........}\\
\end{array}\]
Write the IUPAC name of the following compound:
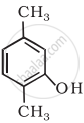
- Draw the structures of all isomeric alcohols of molecular formula C5H12O and give their IUPAC names.
- Classify the isomers of alcohols in the above question as primary, secondary and tertiary alcohols.
Give the IUPAC name of the following ether:
O2N – C6H4 – OCH3(p)
Give the IUPAC name of the following ether:

Natalite is a mixture of
(a) diethyl ether and methanol
(b) diethyl ether and ethanol
(c) dimethyl ether and methanol
(d) dimethyl ether and ethanol
Propanoic acid to ethylamine.
Write IUPAC names of the following

An example of a compound with functional group – O – is ____________.
Butane-2-ol is ____________.
Ethyl alcohol is industrially prepared from ethylene by:
Which of the following compounds is oxidised to prepare methyl ethyl ketone?
1-Propanol and 2-propanol can be best distinguished by:
IUPAC name of the compound \[\begin{array}{cc}
\ce{CH3 - CH - OCH3}\\
\phantom{}|\phantom{....}\\
\phantom{}\ce{CH3}\phantom{..}
\end{array}\] is ______.
Which of the following compounds will react with sodium hydroxide solution in water?
Write steps to carry out the conversion of phenol to aspirin.
Match the starting materials given in Column I with the products formed by these (Column II) in the reaction with HI.
| Column I | Column II | ||
| (i) | CH3—O—CH3 | (a) |  |
| (ii) | \[\begin{array}{cc} \ce{CH3}\phantom{..................}\\ \backslash\phantom{.............}\\ \ce{CH-O-CH3}\\ /\phantom{..............}\\ \ce{CH3}\phantom{..................} \end{array}\] |
(b) | \[\begin{array}{cc} \ce{CH3}\phantom{....}\\ |\phantom{.......}\\ \ce{CH3-C-I + CH3OH}\\ |\phantom{.......}\\ \ce{CH3}\phantom{....} \end{array}\] |
| (iii) | \[\begin{array}{cc} \ce{CH3}\phantom{.}\\ |\phantom{....}\\ \ce{H3C-C-O-CH3}\\ |\phantom{....}\\ \ce{CH3}\phantom{..} \end{array}\] |
(c) | 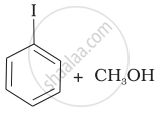 |
| (iv) |  |
(d) | CH3—OH + CH3—I |
| (e) | \[\begin{array}{cc} \ce{CH3}\phantom{.....................}\\ \backslash\phantom{.................}\\ \ce{CH-OH + CH3I}\\ /\phantom{.................}\\ \ce{CH3}\phantom{.....................} \end{array}\] |
||
| (f) | \[\begin{array}{cc} \ce{CH3}\phantom{.....................}\\ \backslash\phantom{.................}\\ \ce{CH-I + CH3OH}\\ /\phantom{.................}\\ \ce{CH3}\phantom{.....................} \end{array}\] |
||
| (g) | \[\begin{array}{cc} \ce{CH3}\phantom{....}\\ |\phantom{.......}\\ \ce{CH3-C-OH + CH3I}\\ |\phantom{.......}\\ \ce{CH3}\phantom{....} \end{array}\] |
Assertion: Like bromination of benzene, bromination of phenol is also carried out in the presence of Lewis acid.
Reason: Lewis acid polarises the bromine molecule.
Assertion: Phenol forms 2, 4, 6 – tribromophenol on treatment with \[\ce{Br2}\] in carbon disulphide at 273 K.
Reason: Bromine polarises in carbon disulphide.
Write the IUPAC name of the following compound.

