Advertisements
Advertisements
प्रश्न
How is phenol converted into the following?
benzoquinone
Write the chemical reaction to convert phenol to benzoquinone.
Advertisements
उत्तर
Phenol on oxidation with Na2Cr2O7 (chromic acid) in H2SO4, gives conjugated diketone known as benzoquinone, which is dark coloured.

संबंधित प्रश्न
Explain metamerism with suitable examples of ethers
Name the following compound according to the IUPAC system.
\[\begin{array}{cc}
\phantom{..................}\ce{CH2OH}\\
\phantom{.........}|\\
\ce{CH3 - CH2 - CH - CH - CH - CH3}\\
\phantom{.........}|\phantom{...................}|\\
\phantom{.............}\ce{CH2Cl}\phantom{..........}\ce{CH3}\phantom{}
\end{array}\]
Name the following compound according to the IUPAC system.
\[\begin{array}{cc}
\ce{CH3 - C = C - CH2OH}\\
|\phantom{......}|\phantom{.....}\\
\ce{CH3}\phantom{.}\ce{Br}\phantom{....}\\
\end{array}\]
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{H3C - CH - CH2 - CH - CH - CH2 - CH3}\\
|\phantom{....................}|\phantom{.........}|\phantom{.............}\\
\ce{OH}\phantom{................}\ce{OH}\phantom{...}\ce{C{_2}H5}\phantom{.........}\\
\end{array}\]
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{CH3 - CH - CH - CH3}\\
|\phantom{.........}|\phantom{...}\\
\ce{OH}\phantom{....}\ce{OH}\\
\end{array}\]
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{HO - CH2 - CH - CH2 - OH}\\
|\phantom{...}\\
\ce{OH}\phantom{.}\\
\end{array}\]
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{CH3 - CH2 - O - CH - CH2 - CH3}\\
\phantom{....}|\\
\phantom{........}\ce{CH3}
\end{array}\]
- Draw the structures of all isomeric alcohols of molecular formula C5H12O and give their IUPAC names.
- Classify the isomers of alcohols in the above question as primary, secondary and tertiary alcohols.
Give the IUPAC name of the following ether:
O2N – C6H4 – OCH3(p)
Write the structure and IUPAC name of 'methyl-n-propyl ether'.
What is the action of hot HI on it?
Give reasons Fluoride ion has higher hydration enthalpy than chloride ion.
In the dehydration of alcohols to alkenes by heating with concentrated sulphuric acid, the initiation step is:
(1) formation of carbonation
(2) formation of an ester
(3) protonation of the alcohol molecule
(4) elimination of water
What.will be the product fonned when chlorobenzene is heated with sodium metal in the presence of dry ether?
Give IUPAC names of the following compound:

The major product formed by the reaction:
\[\begin{array}{cc}
\ce{CH3CH-CH2Br ->[CH3O^-][CH3OH] is}\\
|\phantom{................}\\
\ce{CH3}\phantom{.............}
\end{array}\]
\[\ce{HC ≡ CH ->[HgSO4][H2SO4] ->[CH3MgBr][H2O] ->[PBr3]}\]
What happens when benzene diazonium chloride is heated with water?
Explain why p-nitrophenol is more acidic than phenol.
Match the structures of the compounds given in Column I with the name of the compounds given in Column II.
| Column I | Column II | |
| (i) |  |
(a) Hydroquinone |
| (ii) |  |
(b) Phenetole |
| (iii) | 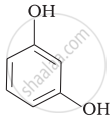 |
(c) Catechol |
| (iv) |  |
(d) o-Cresol |
| (v) |  |
(e) guinone |
| (vi) | 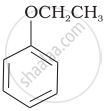 |
(f) Resorcinol |
| (g) Anisole |
Assertion: Addition reaction of water to but-1-ene in acidic medium yields butan-1-ol.
Reason: Addition of water in acidic medium proceeds through the formation of primary carbocation.
Assertion: p-nitrophenol is more acidic than phenol.
Reason: Nitro group helps in the stabilisation of the phenoxide ion by dispersal of negative charge due to resonance.
Assertion: IUPAC name of the compound
\[\begin{array}{cc}
\ce{CH3 - CH - O - CH2 - CH2 - CH3}\\
|\phantom{....................}\\
\ce{CH3}\phantom{.................}
\end{array}\] is 2-Ethoxy-2-methylethane.
Reason: In IUPAC nomenclature, ether is regarded as hydrocarbon derivative in which a hydrogen atom is replaced by —OR or —OAr group [where R = alkyl group and Ar = aryl group]
Assertion: Phenol forms 2, 4, 6 – tribromophenol on treatment with \[\ce{Br2}\] in carbon disulphide at 273 K.
Reason: Bromine polarises in carbon disulphide.
Explain why Lewis acid is not required in bromination of phenol?
How can phenol be converted to aspirin?
Convert the following:
Ethyl alcohol into ethyl acetate.
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{................}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3}\phantom{..}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
Write structural formulae for:
Salicylic acid
