Advertisements
Advertisements
प्रश्न
Give reasons Fluoride ion has higher hydration enthalpy than chloride ion.
Advertisements
उत्तर
Fluoride ion is smaller in size than the chloride ion. Hence, when the two are dissolved in water, the energy of hydration released in the case of fluoride ion will be more than chloride ion due to stronger interactions (ion-dipole) between the ion and the water molecules in the former. Fluorine being more electronegative than chlorine will form stronger H-bonds with hydrogen present in the water molecule.
`triangleH_("hyd")F^(-) > triangle H_("hyd") Cl^(-)`
संबंधित प्रश्न
Name the following compound according to the IUPAC system.
\[\begin{array}{cc}
\ce{CH3 - C = C - CH2OH}\\
|\phantom{......}|\phantom{.....}\\
\ce{CH3}\phantom{.}\ce{Br}\phantom{....}\\
\end{array}\]
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{........................}\ce{CH3}\\
\phantom{....................}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{.........}|\phantom{.........}|\\
\phantom{...}\ce{CH3}\phantom{....}\ce{OH}\phantom{....}\ce{CH3}\\
\end{array}\]
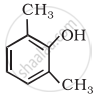
Write the IUPAC name of the following compound:
Natalite is a mixture of
(a) diethyl ether and methanol
(b) diethyl ether and ethanol
(c) dimethyl ether and methanol
(d) dimethyl ether and ethanol
How is phenol converted into the following?
benzoquinone
How is phenol converted into the following?
picric acid
How do you convert the Ethanal to Propanone
Isopropyl alcohol on oxidation forms:
Assertion: Like bromination of benzene, bromination of phenol is also carried out in the presence of Lewis acid.
Reason: Lewis acid polarises the bromine molecule.
Write structural formulae for:
Salicylic acid
