Advertisements
Advertisements
प्रश्न
Give reasons Fluoride ion has higher hydration enthalpy than chloride ion.
Advertisements
उत्तर
Fluoride ion is smaller in size than the chloride ion. Hence, when the two are dissolved in water, the energy of hydration released in the case of fluoride ion will be more than chloride ion due to stronger interactions (ion-dipole) between the ion and the water molecules in the former. Fluorine being more electronegative than chlorine will form stronger H-bonds with hydrogen present in the water molecule.
`triangleH_("hyd")F^(-) > triangle H_("hyd") Cl^(-)`
संबंधित प्रश्न
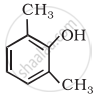
Write the IUPAC name of the following compound:
Ethylidene dichloride when boiled with aqueous solution of NaOH yields _______.
(A) formaldehyde
(B) acetaldehyde
(C) acetone
(D) ethyl methyl ketone
Write the IUPAC name of the following :

How do you convert the Ethanal to Propanone
The compound HOCH2 – CH2OH is __________.
HBr reacts fastest with ____________.
Among the following sets of reactants which one produces anisole?
Assertion: Phenol forms 2, 4, 6 – tribromophenol on treatment with \[\ce{Br2}\] in carbon disulphide at 273 K.
Reason: Bromine polarises in carbon disulphide.
Give the structures of Thiosulphuric acid and Peroxy monosulphuric acid.
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{...............}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3}\phantom{..}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
