Advertisements
Advertisements
प्रश्न
Explain the following terms with suitable examples - Anionic detergents
Define the following: Anionic detergents
Advertisements
उत्तर १
Anionic detergents
Anionic detergents are of two types:
1.Sodium alkyl sulphates: These detergents are sodium salts of long chain alcohols. They are prepared by first treating these alcohols with concentrated sulphuric acid and then with sodium hydroxide. Examples of these detergents include sodium lauryl sulphate (C11H23CH2OSO3−Na+) and sodium stearyl sulphate (C17H35CH2OSO3−Na+).
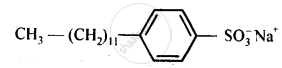
2.Sodium alkylbenzenesulphonates: These detergents are sodium salts of long chain alkylbenzenesulphonic acids. They are prepared by Friedel-Crafts alkylation of benzene with long chain alkyl halides or alkenes. The obtained product is first treated with concentrated sulphuric acid and then with sodium hydroxide. Sodium 4-(1-dodecy) benzenesulphonate (SDS) is an example of anionic detergents.
उत्तर २
Anionic detergents are called so because a large part of their molecules are anions. ‘These are of two types:
(a)Sodium alkyl sulphates: For example, sodium lauryl sulphate, C11H23CH2OSO3 Na+.
(b)Sodium alkylbenzenesulphonates.Vor example, sodium 4-(l-dodecyl) benzenesu Iphphonate (SDS).