Advertisements
Advertisements
प्रश्न
Assertion: p-nitrophenol is more acidic than phenol.
Reason: Nitro group helps in the stabilisation of the phenoxide ion by dispersal of negative charge due to resonance.
पर्याय
Assertion and reason both are correct and reason is correct explanation of assertion.
Assertion and reason both are wrong statements.
Assertion is correct statement but reason is wrong statement.
Assertion is wrong statement but reason is correct statement.
Both assertion and reason are correct statements but reason is not correct explanation of assertion.
Advertisements
उत्तर
Assertion and reason both are correct and reason is correct explanation of assertion.
Explanation:
P-nitro phenol is more acidic as nitro group helps in the stabilization of the phenoxide ion by dispersal of negative charge due to resonance.
APPEARS IN
संबंधित प्रश्न
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{CH3 - O - CH2 - CH - CH3}\\
\phantom{................}|\\
\phantom{....................}\ce{CH3}
\end{array}\]
Write the IUPAC name of the following compound:
C6H5 – O – C2H5
Give the IUPAC name of the following ether:
CH3CH2CH2OCH3
Give the IUPAC name of the following ether:

Which of the following compounds is NOT prepared by the action of alcoholic NI3 on alkyl halide?
(a) CH3NH2
(b) CH3- CH2- NH2
(c) CH3 - CH2 - CH2 - NH2
(d) (CH3)3 C- NH2
How is phenol converted into the following?
Benzene
Write IUPAC name of the following compound (CH3)2 N − CH2CH3
Give reasons Fluoride ion has higher hydration enthalpy than chloride ion.
Write the IUPAC name of the following :

What.will be the product fonned when chlorobenzene is heated with sodium metal in the presence of dry ether?
Write structural formulae for 3-Methoxyhexane
Write structural formulae for Cyclohex-2-en-1-ol.
Ethyl alcohol is industrially prepared from ethylene by:
The correct acidic strength order of the following is:

(I)
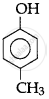
(II)

(III)
Which of the following reagents can be used to oxidise primary alcohols to aldehydes?
(i) \[\ce{CrO3}\] in anhydrous medium.
(ii) \[\ce{KMnO4}\] in acidic medium.
(iii) Pyridinium chlorochromate.
(iv) Heat in the presence of Cu at 573 K.
What happens when benzene diazonium chloride is heated with water?
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{................}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3}\phantom{..}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
Write the IUPAC name.
\[\begin{array}{cc}
\phantom{................}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C -CH3}\\
\phantom{.}|\phantom{......}|\phantom{......}|\\
\phantom{....}\ce{CH3\phantom{...}\ce{OH}\phantom{...}\ce{CH3}}\
\end{array}\]
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{...............}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3}\phantom{..}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
