Advertisements
Advertisements
प्रश्न
Write steps to carry out the conversion of phenol to aspirin.
Advertisements
उत्तर
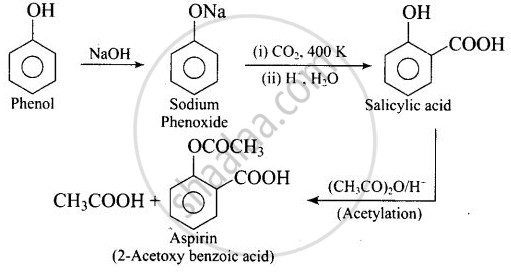
APPEARS IN
संबंधित प्रश्न
Explain metamerism with suitable examples of ethers
Write the IUPAC name of the given compound:

Write the IUPAC name of the following compound:
C6H5 – O – C2H5
- Draw the structures of all isomeric alcohols of molecular formula C5H12O and give their IUPAC names.
- Classify the isomers of alcohols in the above question as primary, secondary and tertiary alcohols.
Give the IUPAC name of the following ether:
CH3OCH2CH2Cl
Give the IUPAC name of the following ether:
O2N – C6H4 – OCH3(p)
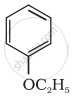
Give the IUPAC name of the following ether:
Ethylidene dichloride when boiled with aqueous solution of NaOH yields _______.
(A) formaldehyde
(B) acetaldehyde
(C) acetone
(D) ethyl methyl ketone
3-Methylbutane-2-ol on heating with HI gives ______
Write the structures of the products when Butan-2-ol reacts with SOCl2
Write IUPAC name of the following

Isopropyl alcohol on oxidation forms:
3-methylphenol is called ____________.
One of the following is not a dihydroxy derivative of benzene.
Which of the following compounds is oxidised to prepare methyl ethyl ketone?
n-Propyl alcohol and isopropyl alcohol can be chemically distinguished by which reagent?
Which of the following compounds will react with sodium hydroxide solution in water?
Arrange the following compounds in decreasing order of acidity.
\[\ce{H2O, ROH, HC ≡ CH}\]
Assertion: Phenol forms 2, 4, 6 – tribromophenol on treatment with \[\ce{Br2}\] in carbon disulphide at 273 K.
Reason: Bromine polarises in carbon disulphide.
