Advertisements
Advertisements
प्रश्न
Write structural formula for pentane-1,4-diol.
Advertisements
उत्तर
\[\begin{array}{cc}
\ce{H3C - HC - H2C - H2C - CH2 - OH}\\
|\phantom{......................}\\
\ce{\underset{Pentane-1,4-diol}{OH}\phantom{....................}}\\
\end{array}\]
संबंधित प्रश्न
Write the IUPAC name of the following compound:

Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{CH3 - CH2 - O - CH - CH2 - CH3}\\
\phantom{....}|\\
\phantom{........}\ce{CH3}
\end{array}\]
- Draw the structures of all isomeric alcohols of molecular formula C5H12O and give their IUPAC names.
- Classify the isomers of alcohols in the above question as primary, secondary and tertiary alcohols.
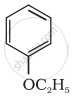
Give the IUPAC name of the following ether:
\[\begin{array}{cc}
\ce{C2H5OCH2 - CH - CH3}\\
\phantom{.........}|\\
\phantom{.............}\ce{CH3}
\end{array}\]
Give the IUPAC name of the following ether:
Ethylidene dichloride when boiled with aqueous solution of NaOH yields _______.
(A) formaldehyde
(B) acetaldehyde
(C) acetone
(D) ethyl methyl ketone
Natalite is a mixture of
(a) diethyl ether and methanol
(b) diethyl ether and ethanol
(c) dimethyl ether and methanol
(d) dimethyl ether and ethanol
How is phenol converted into the following?
Benzene
Write the structures of the products when Butan-2-ol reacts with SOCl2
What.will be the product fonned when chlorobenzene is heated with sodium metal in the presence of dry ether?
Propanoic acid to ethylamine.
Write structural formulae for Cyclohex-2-en-1-ol.
Give IUPAC names of the following compound:

HBr reacts fastest with ____________.
IUPAC name of m-cresol is ____________.
Which of the following gives a positive iodoform test?
The correct acidic strength order of the following is:

(I)
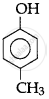
(II)

(III)
Give IUPAC name of the compound given below.
\[\begin{array}{cc}
\phantom{}\ce{CH3 - CH - CH2 - CH2 - CH - CH3}\phantom{.}\\
\phantom{.........}|\phantom{...................}|\phantom{...........}\\
\phantom{..}\ce{Cl}\phantom{.................}\ce{OH}\phantom{..}
\end{array}\]
Match the structures of the compounds given in Column I with the name of the compounds given in Column II.
| Column I | Column II | |
| (i) |  |
(a) Hydroquinone |
| (ii) |  |
(b) Phenetole |
| (iii) | 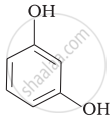 |
(c) Catechol |
| (iv) |  |
(d) o-Cresol |
| (v) |  |
(e) guinone |
| (vi) | 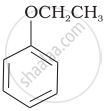 |
(f) Resorcinol |
| (g) Anisole |
Assertion: IUPAC name of the compound
\[\begin{array}{cc}
\ce{CH3 - CH - O - CH2 - CH2 - CH3}\\
|\phantom{....................}\\
\ce{CH3}\phantom{.................}
\end{array}\] is 2-Ethoxy-2-methylethane.
Reason: In IUPAC nomenclature, ether is regarded as hydrocarbon derivative in which a hydrogen atom is replaced by —OR or —OAr group [where R = alkyl group and Ar = aryl group]
Write complete reaction for the bromination of phenol in aqueous and non-aqueous medium.
Write the IUPAC name of the following compound.

Draw structure of the following compound.
Prop-2-en-1-ol
Give the structures of Thiosulphuric acid and Peroxy monosulphuric acid.
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{..............}\ce{CH3}\\
\phantom{............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{.....}|\phantom{......}|\\
\phantom{...}\ce{CH3}\phantom{.}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
Write the IUPAC name.
\[\begin{array}{cc}
\phantom{................}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C -CH3}\\
\phantom{.}|\phantom{......}|\phantom{......}|\\
\phantom{....}\ce{CH3\phantom{...}\ce{OH}\phantom{...}\ce{CH3}}\
\end{array}\]
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{................}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3}\phantom{...}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
