Advertisements
Advertisements
प्रश्न
Write complete reaction for the bromination of phenol in aqueous and non-aqueous medium.
Advertisements
उत्तर
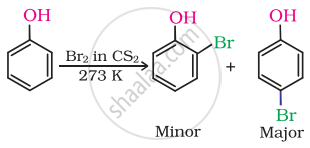
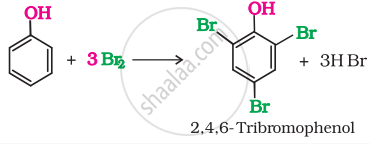
When phenol is treated with bromine water. 2, 4, 6-tribromophenol is formed as while precipitate:
APPEARS IN
संबंधित प्रश्न
Give the IUPAC name of the following ether:
CH3OCH2CH2Cl
Give the IUPAC name of the following ether:
O2N – C6H4 – OCH3(p)
Give reasons Fluoride ion has higher hydration enthalpy than chloride ion.
In the dehydration of alcohols to alkenes by heating with concentrated sulphuric acid, the initiation step is:
(1) formation of carbonation
(2) formation of an ester
(3) protonation of the alcohol molecule
(4) elimination of water
Write IUPAC name of the following
\[\begin{array}{cc}\ce{CH3-CH-CH-CH2-OH}\\|\phantom{.....}|\phantom{.......}\\\ce{OH}\phantom{..}\ce{CH3}\phantom{.....}\end{array}\]
C6H5OCH2CH3 is called:
One of the following is not a dihydroxy derivative of benzene.
Cresol has ____________.
The product of acid catalysed hydration of 2-phenylpropene is:
Which of the following gives a positive iodoform test?
IUPAC name of the compound \[\begin{array}{cc}
\ce{CH3 - CH - OCH3}\\
\phantom{}|\phantom{....}\\
\phantom{}\ce{CH3}\phantom{..}
\end{array}\] is ______.
Which of the following compounds will react with sodium hydroxide solution in water?
What happens when benzene diazonium chloride is heated with water?
Assertion: Like bromination of benzene, bromination of phenol is also carried out in the presence of Lewis acid.
Reason: Lewis acid polarises the bromine molecule.
Explain why Lewis acid is not required in bromination of phenol?
Write the IUPAC name of the following compound.

Convert the following:
Ethyl alcohol into ethyl acetate.
Write a chemical reaction for the following conversion:
Acetic acid into ethyl alcohol.
Draw structure of the following compound.
Prop-2-en-1-ol
The IUPAC name of 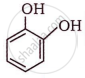 is ______.
is ______.
