Advertisements
Advertisements
प्रश्न
Give IUPAC name of the compound given below.
\[\begin{array}{cc}
\phantom{}\ce{CH3 - CH - CH2 - CH2 - CH - CH3}\phantom{.}\\
\phantom{.........}|\phantom{...................}|\phantom{...........}\\
\phantom{..}\ce{Cl}\phantom{.................}\ce{OH}\phantom{..}
\end{array}\]
पर्याय
2-Chloro-5-hydroxyhexane
2-Hydroxy-5-chlorohexane
5-Chlorohexan-2-ol
2-Chlorohexan-5-ol
Advertisements
उत्तर
5-Chlorohexan-2-ol
Explanation:
\[\begin{array}{cc}
\ce{\overset{6}{C}H3-\overset{5}{C}H-\overset{4}{C}H2-\overset{3}{C}H2-\overset{2}{C}H-\overset{1}{C}H3}\\
\phantom{}|\phantom{.................}|\phantom{..}\\
\phantom{....}\ce{\underset{5-Chlorohexan-2-ol}{Cl\phantom{...............}OH}}\phantom{....}
\end{array}\]
APPEARS IN
संबंधित प्रश्न
Explain metamerism with suitable examples of ethers
Name the following compound according to IUPAC system.

Write IUPAC name of the following compound:
What is the action of hot HI on it?
Write the structures of the products when Butan-2-ol reacts with SOCl2
Write structural formulae for 1-Ethylcyclohexanol.
Write IUPAC name of the following
\[\begin{array}{cc}\ce{CH3-CH-CH-CH2-OH}\\|\phantom{.....}|\phantom{.......}\\\ce{OH}\phantom{..}\ce{CH3}\phantom{.....}\end{array}\]
Write IUPAC names of the following

Ethyl alcohol is industrially prepared from ethylene by:
Ethylene reacts with Baeyer’s reagent to give ______.
The major product formed by the reaction:
\[\begin{array}{cc}
\ce{CH3CH-CH2Br ->[CH3O^-][CH3OH] is}\\
|\phantom{................}\\
\ce{CH3}\phantom{.............}
\end{array}\]
Arrange the following compounds in decreasing order of acidity.
\[\ce{H2O, ROH, HC ≡ CH}\]
Match the structures of the compounds given in Column I with the name of the compounds given in Column II.
| Column I | Column II | |
| (i) |  |
(a) Hydroquinone |
| (ii) |  |
(b) Phenetole |
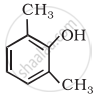
| (iii) | 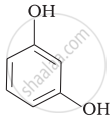 |
(c) Catechol |
| (iv) |  |
(d) o-Cresol |
| (v) |  |
(e) guinone |
| (vi) | 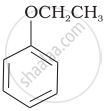 |
(f) Resorcinol |
| (g) Anisole |
Assertion: p-nitrophenol is more acidic than phenol.
Reason: Nitro group helps in the stabilisation of the phenoxide ion by dispersal of negative charge due to resonance.
Write complete reaction for the bromination of phenol in aqueous and non-aqueous medium.
Explain why Lewis acid is not required in bromination of phenol?
How can phenol be converted to aspirin?
Write the IUPAC name of the following compound.

Draw structure of the following compound.
2. 5-Diethylphenol
Draw structure of the following compound.
Prop-2-en-1-ol
