Advertisements
Advertisements
प्रश्न
Give IUPAC name of the compound given below.
\[\begin{array}{cc}
\phantom{}\ce{CH3 - CH - CH2 - CH2 - CH - CH3}\phantom{.}\\
\phantom{.........}|\phantom{...................}|\phantom{...........}\\
\phantom{..}\ce{Cl}\phantom{.................}\ce{OH}\phantom{..}
\end{array}\]
विकल्प
2-Chloro-5-hydroxyhexane
2-Hydroxy-5-chlorohexane
5-Chlorohexan-2-ol
2-Chlorohexan-5-ol
Advertisements
उत्तर
5-Chlorohexan-2-ol
Explanation:
\[\begin{array}{cc}
\ce{\overset{6}{C}H3-\overset{5}{C}H-\overset{4}{C}H2-\overset{3}{C}H2-\overset{2}{C}H-\overset{1}{C}H3}\\
\phantom{}|\phantom{.................}|\phantom{..}\\
\phantom{....}\ce{\underset{5-Chlorohexan-2-ol}{Cl\phantom{...............}OH}}\phantom{....}
\end{array}\]
APPEARS IN
संबंधित प्रश्न
Write IUPAC name of the following compound:
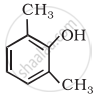
Write IUPAC name of the following compound:
C6H5 – O – C7H15(n−)
Give IUPAC name of the following ether:
\[\begin{array}{cc}
\ce{C2H5OCH2 - CH - CH3}\\
\phantom{.....}|\\
\phantom{.......}\ce{CH3}
\end{array}\]
Give reasons Fluoride ion has higher hydration enthalpy than chloride ion.
Write the IUPAC name of the following compound:

Write the structures of the products when Butan-2-ol reacts with CrO3
Write IUPAC name of the following

Glycerol is ____________.
C6H5OCH2CH3 is called:
Ethylene reacts with Baeyer’s reagent to give ______.
Which of the following is most acidic?
The IUPAC name of the ether CH2 = CH–CH2OCH3 is:
The major product formed by the reaction:
\[\begin{array}{cc}
\ce{CH3CH-CH2Br ->[CH3O^-][CH3OH] is}\\
|\phantom{................}\\
\ce{CH3}\phantom{.............}
\end{array}\]
\[\ce{Phenol ->[Zn, dust] 'X' ->[CH3Cl][Anhy. AlCl3] 'Y' ->[Alkaline][KMnO4] 'Z'}\]
The product ‘Z’ is:
Assertion: Addition reaction of water to but-1-ene in acidic medium yields butan-1-ol.
Reason: Addition of water in acidic medium proceeds through the formation of primary carbocation.
Write a chemical reaction for the following conversion:
Acetic acid into ethyl alcohol.
Draw structure of the following compound.
2. 5-Diethylphenol
Give the structures of Thiosulphuric acid and Peroxy monosulphuric acid.
Write structural formulae for:
Salicylic acid
