Advertisements
Advertisements
प्रश्न
Write IUPAC name of the following

Advertisements
उत्तर
1-Methylcyclopentanol
APPEARS IN
संबंधित प्रश्न
Explain metamerism with suitable examples of ethers
Write the IUPAC name of the given compound:

Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{H3C - CH - CH2 - CH - CH - CH2 - CH3}\\
|\phantom{....................}|\phantom{.........}|\phantom{.............}\\
\ce{OH}\phantom{................}\ce{OH}\phantom{...}\ce{C{_2}H5}\phantom{.........}\\
\end{array}\]
- Draw the structures of all isomeric alcohols of molecular formula C5H12O and give their IUPAC names.
- Classify the isomers of alcohols in the above question as primary, secondary and tertiary alcohols.
Give the IUPAC name of the following ether:

What is the action of hot HI on it?
Give reasons Fluoride ion has higher hydration enthalpy than chloride ion.
Write the IUPAC name of the following compound:

Propanoic acid to ethylamine.
Write structural formula for pentane-1,4-diol.
Write structural formulae for Cyclohex-2-en-1-ol.
3-methylphenol is called ____________.
When ethyl alcohol reacts with acetic acid, the products formed are:
The product of acid catalysed hydration of 2-phenylpropene is:
\[\ce{Phenol ->[Zn, dust] 'X' ->[CH3Cl][Anhy. AlCl3] 'Y' ->[Alkaline][KMnO4] 'Z'}\]
The product ‘Z’ is:
Give IUPAC name of the compound given below.
\[\begin{array}{cc}
\phantom{}\ce{CH3 - CH - CH2 - CH2 - CH - CH3}\phantom{.}\\
\phantom{.........}|\phantom{...................}|\phantom{...........}\\
\phantom{..}\ce{Cl}\phantom{.................}\ce{OH}\phantom{..}
\end{array}\]
Write steps to carry out the conversion of phenol to aspirin.
Match the structures of the compounds given in Column I with the name of the compounds given in Column II.
| Column I | Column II | |
| (i) |  |
(a) Hydroquinone |
| (ii) |  |
(b) Phenetole |
| (iii) | 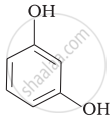 |
(c) Catechol |
| (iv) |  |
(d) o-Cresol |
| (v) |  |
(e) guinone |
| (vi) | 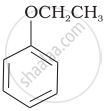 |
(f) Resorcinol |
| (g) Anisole |
Assertion: Phenol forms 2, 4, 6 – tribromophenol on treatment with \[\ce{Br2}\] in carbon disulphide at 273 K.
Reason: Bromine polarises in carbon disulphide.
Convert the following:
Ethyl alcohol into ethyl acetate.
Write a chemical reaction for the following conversion:
Acetic acid into ethyl alcohol.
How are the following conversions carried out?
Methyl magnesium bromide→2-Methylpropan-2-ol.
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{................}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3}\phantom{..}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
Draw structure of the following compound.
Prop-2-en-1-ol
Write structural formulae for:
Salicylic acid
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{................}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3}\phantom{...}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{...............}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3}\phantom{..}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
