Advertisements
Advertisements
प्रश्न
Write IUPAC name of the following
\[\begin{array}{cc}\ce{CH3-CH-CH-CH2-OH}\\|\phantom{.....}|\phantom{.......}\\\ce{OH}\phantom{..}\ce{CH3}\phantom{.....}\end{array}\]
Advertisements
उत्तर
2-Methylbutane-1,3-diol
APPEARS IN
संबंधित प्रश्न
Write the IUPAC name of the following compound:
C6H5 – O – C7H15(n−)
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{CH3 - CH2 - O - CH - CH2 - CH3}\\
\phantom{....}|\\
\phantom{........}\ce{CH3}
\end{array}\]
Write structures of the compounds whose IUPAC names are as follows:
3-Chloromethylpentan-1-ol.
How is phenol converted into the following?
Benzene
Write IUPAC name of the following compound (CH3)2 N − CH2CH3
Write IUPAC name of the following

Give IUPAC names of the following compound:

3-methylphenol is called ____________.
The compound HOCH2 – CH2OH is __________.
An example of a compound with functional group – O – is ____________.
HBr reacts fastest with ____________.
1-Propanol and 2-propanol can be best distinguished by:
The heating of phenyl methyl ether with HI produces:
The correct acidic strength order of the following is:

(I)
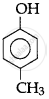
(II)

(III)
Write the IUPAC name of the compound given below.
\[\begin{array}{cc}
\phantom{}\ce{CH3 - CH2 - C = C - OH}\\
\phantom{........}|\phantom{....}|\phantom{}\\
\phantom{..............}\ce{CH3 CH2OH}
\end{array}\]
What happens when benzene diazonium chloride is heated with water?
Arrange the following compounds in decreasing order of acidity.
\[\ce{H2O, ROH, HC ≡ CH}\]
Write steps to carry out the conversion of phenol to aspirin.
Assertion: p-nitrophenol is more acidic than phenol.
Reason: Nitro group helps in the stabilisation of the phenoxide ion by dispersal of negative charge due to resonance.
Assertion: Phenol forms 2, 4, 6 – tribromophenol on treatment with \[\ce{Br2}\] in carbon disulphide at 273 K.
Reason: Bromine polarises in carbon disulphide.
How can phenol be converted to aspirin?
Write the IUPAC name of the following compound.

Identify A and B in the following:

Write structural formulae for:
p-Nitrophenol
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{...............}\ce{CH3}\\
\phantom{............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3}\phantom{...}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
