Advertisements
Advertisements
प्रश्न
What happens when benzene diazonium chloride is heated with water?
Advertisements
उत्तर
When benzene diazonium chloride is heated with water, it forms phenol along with nitrogen and hydrochloric acid as by-products as shown below:
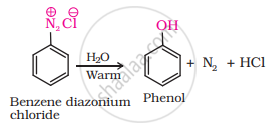
APPEARS IN
संबंधित प्रश्न
Name the following compound according to the IUPAC system.
\[\begin{array}{cc}
\phantom{..................}\ce{CH2OH}\\
\phantom{.........}|\\
\ce{CH3 - CH2 - CH - CH - CH - CH3}\\
\phantom{.........}|\phantom{...................}|\\
\phantom{.............}\ce{CH2Cl}\phantom{..........}\ce{CH3}\phantom{}
\end{array}\]
Name the following compound according to the IUPAC system.

Name the following compound according to the IUPAC system.
\[\begin{array}{cc}
\ce{CH3 - C = C - CH2OH}\\
|\phantom{......}|\phantom{.....}\\
\ce{CH3}\phantom{.}\ce{Br}\phantom{....}\\
\end{array}\]
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{........................}\ce{CH3}\\
\phantom{....................}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{.........}|\phantom{.........}|\\
\phantom{...}\ce{CH3}\phantom{....}\ce{OH}\phantom{....}\ce{CH3}\\
\end{array}\]
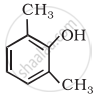
Write the IUPAC name of the following compound:
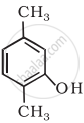
Write the IUPAC name of the following compound:
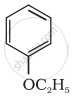
Give the IUPAC name of the following ether:
What is the action of hot HI on it?
In a carbinol system of nomenclature tert.butyl alcohol is named as _______________
n-Propyl alcohol and isopropyl alcohol can be chemically distinguished by which reagent?
Ethylene reacts with Baeyer’s reagent to give ______.
The major product formed by the reaction:
\[\begin{array}{cc}
\ce{CH3CH-CH2Br ->[CH3O^-][CH3OH] is}\\
|\phantom{................}\\
\ce{CH3}\phantom{.............}
\end{array}\]
The product of acid catalysed hydration of 2-phenylpropene is:
The heating of phenyl methyl ether with HI produces:
\[\ce{Phenol ->[Zn, dust] 'X' ->[CH3Cl][Anhy. AlCl3] 'Y' ->[Alkaline][KMnO4] 'Z'}\]
The product ‘Z’ is:
Identify A and B in the following:

Draw structure of the following compound.
2-Methoxypropane
Write structural formulae for:
Salicylic acid
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{................}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3}\phantom{...}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
