Advertisements
Advertisements
प्रश्न
What is the action of hot HI on it?
Advertisements
उत्तर
Action of hot HI on methyl n-propyl ether:
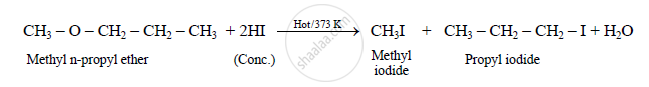
APPEARS IN
संबंधित प्रश्न
What is metamerism?
Name the following compound according to the IUPAC system.
\[\begin{array}{cc}
\phantom{..................}\ce{CH2OH}\\
\phantom{.........}|\\
\ce{CH3 - CH2 - CH - CH - CH - CH3}\\
\phantom{.........}|\phantom{...................}|\\
\phantom{.............}\ce{CH2Cl}\phantom{..........}\ce{CH3}\phantom{}
\end{array}\]
Name the following compound according to the IUPAC system.
\[\begin{array}{cc}
\ce{H2C = CH - CH - CH2 - CH2 - CH3}\\
|\phantom{...............}\\
\ce{OH}\phantom{............}
\end{array}\]
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{HO - CH2 - CH - CH2 - OH}\\
|\phantom{...}\\
\ce{OH}\phantom{.}\\
\end{array}\]
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{CH3 - CH2 - O - CH - CH2 - CH3}\\
\phantom{....}|\\
\phantom{........}\ce{CH3}
\end{array}\]
Give the IUPAC name of the following ether:

Natalite is a mixture of
(a) diethyl ether and methanol
(b) diethyl ether and ethanol
(c) dimethyl ether and methanol
(d) dimethyl ether and ethanol
Propanoic acid to ethylamine.
Write IUPAC names of the following

In a carbinol system of nomenclature tert.butyl alcohol is named as _______________
Isopropyl alcohol on oxidation forms:
Give IUPAC names of the following compound:
\[\begin{array}{cc}
\phantom{..}\ce{H}\phantom{...}\ce{CH3}\phantom{.}\ce{H}\phantom{..}\\
\phantom{}|\phantom{....}|\phantom{....}|\phantom{}\\
\ce{H - C - C - C - H}\\
\phantom{}|\phantom{....}|\phantom{....}|\phantom{}\\
\phantom{.}\ce{H}\phantom{...}\ce{OH}\phantom{.}\ce{H}\phantom{.}\\
\end{array}\]
An example of a compound with functional group – O – is ____________.
Butane-2-ol is ____________.
Cresol has ____________.
Among the following sets of reactants which one produces anisole?
Write the IUPAC name of the compound given below.
\[\begin{array}{cc}
\phantom{}\ce{CH3 - CH2 - C = C - OH}\\
\phantom{........}|\phantom{....}|\phantom{}\\
\phantom{..............}\ce{CH3 CH2OH}
\end{array}\]
Arrange the following compounds in decreasing order of acidity.
\[\ce{H2O, ROH, HC ≡ CH}\]
Match the starting materials given in Column I with the products formed by these (Column II) in the reaction with HI.
| Column I | Column II | ||
| (i) | CH3—O—CH3 | (a) |  |
| (ii) | \[\begin{array}{cc} \ce{CH3}\phantom{..................}\\ \backslash\phantom{.............}\\ \ce{CH-O-CH3}\\ /\phantom{..............}\\ \ce{CH3}\phantom{..................} \end{array}\] |
(b) | \[\begin{array}{cc} \ce{CH3}\phantom{....}\\ |\phantom{.......}\\ \ce{CH3-C-I + CH3OH}\\ |\phantom{.......}\\ \ce{CH3}\phantom{....} \end{array}\] |
| (iii) | \[\begin{array}{cc} \ce{CH3}\phantom{.}\\ |\phantom{....}\\ \ce{H3C-C-O-CH3}\\ |\phantom{....}\\ \ce{CH3}\phantom{..} \end{array}\] |
(c) | 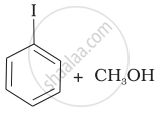 |
| (iv) |  |
(d) | CH3—OH + CH3—I |
| (e) | \[\begin{array}{cc} \ce{CH3}\phantom{.....................}\\ \backslash\phantom{.................}\\ \ce{CH-OH + CH3I}\\ /\phantom{.................}\\ \ce{CH3}\phantom{.....................} \end{array}\] |
||
| (f) | \[\begin{array}{cc} \ce{CH3}\phantom{.....................}\\ \backslash\phantom{.................}\\ \ce{CH-I + CH3OH}\\ /\phantom{.................}\\ \ce{CH3}\phantom{.....................} \end{array}\] |
||
| (g) | \[\begin{array}{cc} \ce{CH3}\phantom{....}\\ |\phantom{.......}\\ \ce{CH3-C-OH + CH3I}\\ |\phantom{.......}\\ \ce{CH3}\phantom{....} \end{array}\] |
Assertion: Like bromination of benzene, bromination of phenol is also carried out in the presence of Lewis acid.
Reason: Lewis acid polarises the bromine molecule.
How can phenol be converted to aspirin?
Write the IUPAC name of the following compound.

Write a chemical reaction for the following conversion:
Acetic acid into ethyl alcohol.
Draw structure of the following compound.
2. 5-Diethylphenol
Give the structures of Thiosulphuric acid and Peroxy monosulphuric acid.
Write structural formulae for:
p-Nitrophenol
Write structural formulae for:
Salicylic acid
Write the IUPAC name.
\[\begin{array}{cc}
\phantom{................}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C -CH3}\\
\phantom{.}|\phantom{......}|\phantom{......}|\\
\phantom{....}\ce{CH3\phantom{...}\ce{OH}\phantom{...}\ce{CH3}}\
\end{array}\]
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{................}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3}\phantom{...}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
