Advertisements
Advertisements
प्रश्न
Write IUPAC names of the following

Advertisements
उत्तर
2-Nitrophenol
APPEARS IN
संबंधित प्रश्न
Name the following compound according to the IUPAC system.
\[\begin{array}{cc}
\ce{H2C = CH - CH - CH2 - CH2 - CH3}\\
|\phantom{...............}\\
\ce{OH}\phantom{............}
\end{array}\]
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{........................}\ce{CH3}\\
\phantom{....................}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{.........}|\phantom{.........}|\\
\phantom{...}\ce{CH3}\phantom{....}\ce{OH}\phantom{....}\ce{CH3}\\
\end{array}\]
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{H3C - CH - CH2 - CH - CH - CH2 - CH3}\\
|\phantom{....................}|\phantom{.........}|\phantom{.............}\\
\ce{OH}\phantom{................}\ce{OH}\phantom{...}\ce{C{_2}H5}\phantom{.........}\\
\end{array}\]
Write the IUPAC name of the following compound:
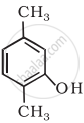
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{CH3 - CH2 - O - CH - CH2 - CH3}\\
\phantom{....}|\\
\phantom{........}\ce{CH3}
\end{array}\]
Write structures of the compounds whose IUPAC names are as follows:
3-Chloromethylpentan-1-ol.
Give the IUPAC name of the following ether:
CH3CH2CH2OCH3
Natalite is a mixture of
(a) diethyl ether and methanol
(b) diethyl ether and ethanol
(c) dimethyl ether and methanol
(d) dimethyl ether and ethanol
How is phenol converted into the following?
picric acid
Write the IUPAC name of the following compound:

What.will be the product fonned when chlorobenzene is heated with sodium metal in the presence of dry ether?
Resorcinol on distillation with zinc dust gives _________.
Write structural formulae for 3-Methoxyhexane
Write structural formula for pentane-1,4-diol.
Give IUPAC names of the following compound:
\[\begin{array}{cc}
\phantom{..}\ce{H}\phantom{...}\ce{CH3}\phantom{.}\ce{H}\phantom{..}\\
\phantom{}|\phantom{....}|\phantom{....}|\phantom{}\\
\ce{H - C - C - C - H}\\
\phantom{}|\phantom{....}|\phantom{....}|\phantom{}\\
\phantom{.}\ce{H}\phantom{...}\ce{OH}\phantom{.}\ce{H}\phantom{.}\\
\end{array}\]
One of the following is not a dihydroxy derivative of benzene.
An example of a compound with functional group – O – is ____________.
Ethyl alcohol is industrially prepared from ethylene by:
HBr reacts fastest with ____________.
Among the following sets of reactants which one produces anisole?
IUPAC name of the compound is:
\[\begin{array}{cc}
\ce{CH3-CH-OCH3}\\
|\phantom{....}\\
\ce{CH3}\phantom{..}
\end{array}\]
What happens when benzene diazonium chloride is heated with water?
Match the structures of the compounds given in Column I with the name of the compounds given in Column II.
| Column I | Column II | |
| (i) |  |
(a) Hydroquinone |
| (ii) |  |
(b) Phenetole |
| (iii) | 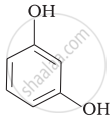 |
(c) Catechol |
| (iv) |  |
(d) o-Cresol |
| (v) |  |
(e) guinone |
| (vi) | 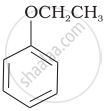 |
(f) Resorcinol |
| (g) Anisole |
Match the starting materials given in Column I with the products formed by these (Column II) in the reaction with HI.
| Column I | Column II | ||
| (i) | CH3—O—CH3 | (a) |  |
| (ii) | \[\begin{array}{cc} \ce{CH3}\phantom{..................}\\ \backslash\phantom{.............}\\ \ce{CH-O-CH3}\\ /\phantom{..............}\\ \ce{CH3}\phantom{..................} \end{array}\] |
(b) | \[\begin{array}{cc} \ce{CH3}\phantom{....}\\ |\phantom{.......}\\ \ce{CH3-C-I + CH3OH}\\ |\phantom{.......}\\ \ce{CH3}\phantom{....} \end{array}\] |
| (iii) | \[\begin{array}{cc} \ce{CH3}\phantom{.}\\ |\phantom{....}\\ \ce{H3C-C-O-CH3}\\ |\phantom{....}\\ \ce{CH3}\phantom{..} \end{array}\] |
(c) | 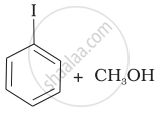 |
| (iv) |  |
(d) | CH3—OH + CH3—I |
| (e) | \[\begin{array}{cc} \ce{CH3}\phantom{.....................}\\ \backslash\phantom{.................}\\ \ce{CH-OH + CH3I}\\ /\phantom{.................}\\ \ce{CH3}\phantom{.....................} \end{array}\] |
||
| (f) | \[\begin{array}{cc} \ce{CH3}\phantom{.....................}\\ \backslash\phantom{.................}\\ \ce{CH-I + CH3OH}\\ /\phantom{.................}\\ \ce{CH3}\phantom{.....................} \end{array}\] |
||
| (g) | \[\begin{array}{cc} \ce{CH3}\phantom{....}\\ |\phantom{.......}\\ \ce{CH3-C-OH + CH3I}\\ |\phantom{.......}\\ \ce{CH3}\phantom{....} \end{array}\] |
Assertion: Addition reaction of water to but-1-ene in acidic medium yields butan-1-ol.
Reason: Addition of water in acidic medium proceeds through the formation of primary carbocation.
Assertion: Phenol forms 2, 4, 6 – tribromophenol on treatment with \[\ce{Br2}\] in carbon disulphide at 273 K.
Reason: Bromine polarises in carbon disulphide.
Assertion: Phenols give o- and p-nitrophenol on nitration with conc. \[\ce{HNO3}\] and \[\ce{H2SO4}\] mixture.
Reason: –OH group in phenol is o–, p– directing.
Explain why Lewis acid is not required in bromination of phenol?
Convert the following:
Ethyl alcohol into ethyl acetate.
How are the following conversions carried out?
Methyl magnesium bromide→2-Methylpropan-2-ol.
