Advertisements
Advertisements
प्रश्न
Write the structures of the products when Butan-2-ol reacts with CrO3
Advertisements
उत्तर
Secondary alcohol (butan-2-ol) is oxidised to ketone (butan-2-one) by chromic anhyride (CrO3).

APPEARS IN
संबंधित प्रश्न
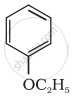
Give the IUPAC name of the following ether:
Write the structure and IUPAC name of 'methyl-n-propyl ether'.
Write IUPAC name of the following compound (CH3)2 N − CH2CH3
Write structural formulae for Cyclohex-2-en-1-ol.
HBr reacts fastest with ____________.
IUPAC name of m-cresol is ____________.
1-Propanol and 2-propanol can be best distinguished by:
\[\ce{HC ≡ CH ->[HgSO4][H2SO4] ->[CH3MgBr][H2O] ->[PBr3]}\]
IUPAC name of the compound \[\begin{array}{cc}
\ce{CH3 - CH - OCH3}\\
\phantom{}|\phantom{....}\\
\phantom{}\ce{CH3}\phantom{..}
\end{array}\] is ______.
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{................}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3}\phantom{...}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
