Advertisements
Advertisements
प्रश्न
Write IUPAC name of the following compound:
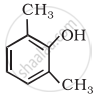
Advertisements
उत्तर
2, 6-Dimethylphenol
APPEARS IN
संबंधित प्रश्न
Name the following compound according to IUPAC system.

Write IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{CH3 - CH2 - O - CH - CH2 - CH3}\\
\phantom{...}|\\
\phantom{.....}\ce{CH3}
\end{array}\]
3-Methylbutane-2-ol on heating with HI gives ______
Write the structures of the products when Butan-2-ol reacts with CrO3
How do you convert the Ethanal to Propanone
What.will be the product fonned when chlorobenzene is heated with sodium metal in the presence of dry ether?
Write IUPAC name of the following

Write IUPAC name of the following
\[\begin{array}{cc}\ce{CH3-CH-CH-CH2-OH}\\|\phantom{.....}|\phantom{.......}\\\ce{OH}\phantom{..}\ce{CH3}\phantom{.....}\end{array}\]
Write IUPAC names of the following

Give IUPAC names of the following compound:

Give IUPAC names of the following compound:
\[\begin{array}{cc}
\phantom{..}\ce{H}\phantom{...}\ce{CH3}\phantom{.}\ce{H}\phantom{..}\\
\phantom{}|\phantom{....}|\phantom{....}|\phantom{}\\
\ce{H - C - C - C - H}\\
\phantom{}|\phantom{....}|\phantom{....}|\phantom{}\\
\phantom{.}\ce{H}\phantom{...}\ce{OH}\phantom{.}\ce{H}\phantom{.}\\
\end{array}\]
The compound HOCH2 – CH2OH is __________.
Ethyl alcohol is industrially prepared from ethylene by:
The correct acidic strength order of the following is:

(I)
(II)

(III)
What happens when benzene diazonium chloride is heated with water?
Write complete reaction for the bromination of phenol in aqueous and non-aqueous medium.
Identify A and B in the following:

The IUPAC name of 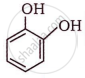 is ______.
is ______.
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{..............}\ce{CH3}\\
\phantom{............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{.....}|\phantom{......}|\\
\phantom{...}\ce{CH3}\phantom{.}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
Write IUPAC names of the following compounds:
\[\begin{array}{cc}
\phantom{...............}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3\phantom{...}OH\phantom{...}CH3}\\
\end{array}\]
