Advertisements
Advertisements
Question
Write the IUPAC name of the following compound:
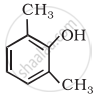
Advertisements
Solution
2,6-Dimethylphenol
APPEARS IN
RELATED QUESTIONS
Write the IUPAC name of the given compound:

Name the following compound according to the IUPAC system.
\[\begin{array}{cc}
\phantom{..................}\ce{CH2OH}\\
\phantom{.........}|\\
\ce{CH3 - CH2 - CH - CH - CH - CH3}\\
\phantom{.........}|\phantom{...................}|\\
\phantom{.............}\ce{CH2Cl}\phantom{..........}\ce{CH3}\phantom{}
\end{array}\]
Name the following compound according to the IUPAC system.

Write IUPAC name of the following compound:

Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{CH3 - O - CH2 - CH - CH3}\\
\phantom{................}|\\
\phantom{....................}\ce{CH3}
\end{array}\]
Write the IUPAC name of the following compound:
C6H5 – O – C2H5
Give IUPAC name of the following ether:
O2N – C6H4 – OCH3(p)
Ethylidene dichloride when boiled with aqueous solution of NaOH yields _______.
(A) formaldehyde
(B) acetaldehyde
(C) acetone
(D) ethyl methyl ketone
Write structural formulae for 1-Ethylcyclohexanol.
Write structural formula for pentane-1,4-diol.
Ethyl alcohol is industrially prepared from ethylene by:
n-Propyl alcohol and isopropyl alcohol can be chemically distinguished by which reagent?
When ethyl alcohol reacts with acetic acid, the products formed are:
The heating of phenyl methyl ether with HI produces:
Which of the following gives a positive iodoform test?
IUPAC name of the compound \[\begin{array}{cc}
\ce{CH3 - CH - OCH3}\\
\phantom{}|\phantom{....}\\
\phantom{}\ce{CH3}\phantom{..}
\end{array}\] is ______.
Write the IUPAC name of the compound given below.
\[\begin{array}{cc}
\phantom{}\ce{CH3 - CH2 - C = C - OH}\\
\phantom{........}|\phantom{....}|\phantom{}\\
\phantom{..............}\ce{CH3 CH2OH}
\end{array}\]
Assertion: Addition reaction of water to but-1-ene in acidic medium yields butan-1-ol.
Reason: Addition of water in acidic medium proceeds through the formation of primary carbocation.
Explain why Lewis acid is not required in bromination of phenol?
